Advanced liver cancer stage 4 treatment in Germany including TACE and dendritic cell therapy with complete logistical support through Treatment in Germany (TIG).
Advanced Liver Cancer Stage 4 Treatment in Germany
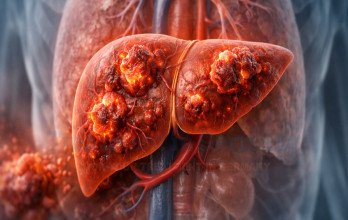
Advanced liver cancer stage 4 refers to hepatocellular carcinoma or secondary liver malignancy that has spread beyond the liver to distant organs or major blood vessels. At this stage, the disease is considered systemic and complex, requiring comprehensive medical planning rather than a single localized therapy. Tumor burden, liver function, and overall physical condition play critical roles in determining the most appropriate management approach. While stage 4 disease is advanced, modern oncology focuses on disease control, symptom reduction, and stabilization of liver function. Understanding structured options for advanced liver cancer stage 4 treatment is essential for informed decision-making.
Germany has become a recognized destination for patients seeking new and innovative treatment options for advanced liver cancer stage 4 treatment in Germany, particularly in complex metastatic cases. Treatment strategies are developed within specialized German healthcare centers and expert-led German oncology centers, where multidisciplinary tumor boards assess imaging findings, tumor biology, and liver reserve before recommending therapy. Interventional oncology procedures are associated with Prof. Vogl, known for advanced image-guided embolization techniques, while immune-based therapies such as dendritic cell treatment are linked to Prof. Gansauge, recognized for his focused work in cellular immunotherapy. Many international patients consult specialists considered among the best oncologist in Germany, drawn by structured treatment pathways and access to latest medical technology in Germany.
Understanding Stage 4 Liver Cancer
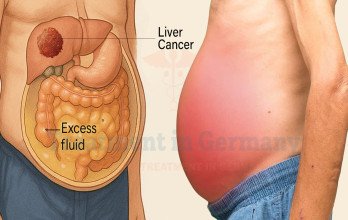
Stage 4 liver cancer indicates that the tumor has either invaded major blood vessels, spread to distant lymph nodes, or metastasized to other organs such as the lungs or bones. Because the liver performs essential metabolic and detoxification functions, maintaining organ performance is a critical component of treatment planning. Patients may present with fatigue, abdominal swelling, jaundice, or unexplained weight loss.
The biological behavior of liver cancer depends on tumor size, number of lesions, and underlying liver health, especially in patients with cirrhosis. Accurate staging determines whether interventional therapies, systemic treatment, or immune-based approaches are appropriate. Comprehensive evaluation is necessary before initiating advanced therapy.
Advanced Diagnostics for Stage 4 Liver Cancer in Germany
Precise staging and liver function assessment are fundamental to therapy selection. In Germany, advanced Diagnostics include biopsy when appropriate, liver function tests, and imaging such as CT scans, MRI, and PET scans to determine tumor distribution and vascular involvement.
These imaging modalities are supported by latest medical technology in Germany, allowing detailed anatomical mapping and treatment planning. Tumor markers and molecular profiling may also influence therapeutic decisions. Findings are reviewed collectively within multidisciplinary tumor boards at German Oncology centers, ensuring coordinated and individualized care.
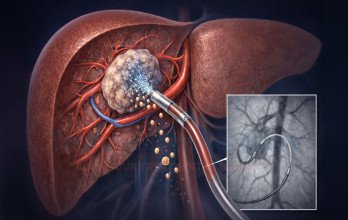
TACE Treatment for Advanced Liver Cancer Stage 4 in Germany
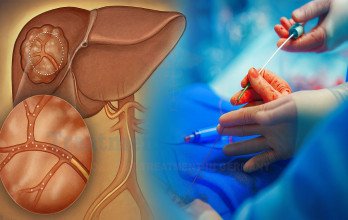
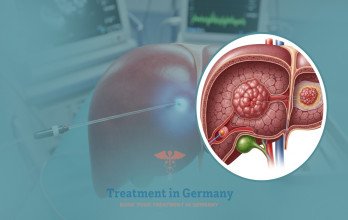
TACE Treatment, or Transarterial Chemoembolization, is an interventional oncology procedure commonly evaluated in advanced liver cancer when localized tumor control is required. Even in stage 4 disease, dominant liver lesions may significantly influence symptoms and overall progression. TACE is designed to deliver concentrated chemotherapy directly into the arteries supplying the tumor while simultaneously reducing its blood supply.
The procedure is performed under real-time imaging guidance in a specialized interventional suite. A catheter is inserted through an artery in the groin and carefully navigated toward the vessels feeding the liver tumor. Chemotherapy is infused directly into the targeted region, followed by embolic particles that block the blood flow to the tumor. This dual mechanism increases local drug concentration while limiting systemic exposure.
Following TACE Treatment, structured follow-up includes liver function monitoring and repeat imaging to evaluate tumor response. Additional sessions may be considered depending on disease behavior and liver reserve. The cost of TACE Treatment in Germany typically ranges between €8,000 and €9,000 per session, depending on clinical complexity and individualized planning.
Immunotherapy: Dendritic Cell Therapy for Advanced Liver Cancer in Germany
Immunotherapy represents an evolving component of advanced liver cancer management. The dendritic cell vaccine approach is designed to stimulate the immune system to recognize tumor-associated antigens expressed by liver cancer cells. Because advanced tumors may suppress immune detection, immune-based therapies aim to enhance systemic immune surveillance and complement interventional or systemic treatment.
The process begins with the collection of immune cells (monocytes) from the patient’s blood under medical supervision. These cells are isolated in a sterile clinical environment to preserve viability and biological integrity. Strict handling protocols ensure optimal quality before laboratory processing.
Collected cells are transferred to an EU GMP certified laboratory, where they are cultured under strictly regulated and quality-controlled conditions. Within this environment, the monocytes are exposed to tumor-associated antigens derived from liver cancer tissue. This controlled exposure promotes maturation into active dendritic cells capable of presenting cancer markers to T-cells. Comprehensive sterility and viability testing is conducted before administration.
Once prepared, the cells are reintroduced into the patient as a personalized immune-based therapy commonly referred to as a dendritic cell vaccine. This therapy is typically integrated alongside TACE or systemic therapy rather than replacing them. Continuous monitoring evaluates immune response and tolerance throughout the treatment course. The cost of Dendritic Cell Therapy in Germany is €27,000, covering immune cell collection, laboratory processing, preparation, and administration.
Combining Modern Treatment Strategies in Germany
Advanced liver cancer stage 4 often requires a multimodal strategy combining interventional procedures, systemic therapy, and supportive care. TACE may help control dominant liver lesions, while immune-based approaches may support systemic disease management.
Treatment planning within German Healthcare centers emphasizes preservation of liver function and symptom stabilization. Continuous reassessment through imaging ensures adaptive therapy adjustments. Multidisciplinary coordination within German Oncology centers allows personalized care aligned with disease progression.
New Clinical Trials for Advanced Liver Cancer Stage 4 in Germany
Patients with treatment-resistant or progressive disease may be evaluated for participation in new clinical trials for advanced Liver Cancer Stage 4 in Germany. These structured research programs investigate emerging immunotherapies and targeted strategies under strict regulatory oversight.
Clinical research standards align with international frameworks referenced by the Canadian Cancer Society, emphasizing patient safety and scientific transparency. Eligibility requires structured evaluation within accredited German oncology centers.
Limitations and Considerations for Advanced Liver Cancer Stage 4 Treatment
Not every patient with advanced liver cancer stage 4 is an appropriate candidate for interventional procedures such as TACE or for immune-based therapies like dendritic cell treatment. Careful medical evaluation by the treating physician is required to assess liver function reserve, extent of tumor involvement, immune status, and overall physical stability. In situations where liver performance is significantly compromised or disease is widely distributed, systemic therapy or supportive care may represent the safest and most suitable primary approach. Treatment recommendations are individualized within specialized German oncology centers to ensure clinical safety and realistic therapeutic goals.
Ongoing monitoring is a central component of advanced liver cancer management. Imaging studies such as CT scans, MRI, and PET scans are performed regularly to evaluate treatment response, identify progression, and guide further decision-making. Laboratory testing and clinical assessments complement imaging to provide a comprehensive overview of liver performance and tumor behavior. The overall strategy focuses on disease stabilization, symptom reduction, and preservation of remaining liver function. Management within German Healthcare centers emphasizes long-term control and quality of life rather than guaranteed cure.
International Patient Support through Treatment in Germany
Access to advanced management for liver cancer involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as Immunotherapy Dendritic cell therapy and TACE therapy, Through Treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This preliminary evaluation by the treating physician helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and Innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:















.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)