Metastatic Stage 4 colorectal cancer survival rate and advanced treatment in Germany including TACE Treatment and Immunotherapy Dendritic cell therapy with coordinated support through TIG.
Metastatic Stage 4 Colorectal Cancer Facts & Survival Rate
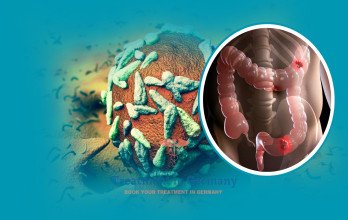
Metastatic Stage 4 colorectal cancer refers to colorectal carcinoma that has progressed beyond the primary tumor site in the colon or rectum and has established secondary tumor deposits in distant organs such as the liver, lungs, or peritoneum. This stage represents systemic disease and requires comprehensive oncologic management. According to Global Cancer Statistics 2020, colorectal cancer accounted for 1,931,590 new cases and 935,173 deaths worldwide in 2020 [1], making it one of the most prevalent and deadly malignancies globally. The burden of disease reflects both incidence and mortality, underscoring the clinical challenge posed by advanced-stage presentation.
Data from the Surveillance, Epidemiology, and End Results (SEER) Program report that the 5-year relative survival rate for distant-stage colorectal cancer is 15% [2]. This survival rate is derived from population-based registry data and reflects outcomes across diverse patient populations, including those with varying comorbidities and treatment access. The relatively low long-term survival emphasizes the biological aggressiveness of metastatic disease and the complexity of achieving durable remission once distant spread has occurred.
Despite these statistics, meaningful progress has been made in systemic therapy over the past two decades. Clinical trials evaluating modern chemotherapy combinations and biologic agents have demonstrated substantial improvements in median overall survival compared with earlier treatment eras [3]. These gains are primarily observed in carefully selected trial populations and highlight the importance of personalized treatment strategies. Management of Metastatic Stage 4 colorectal cancer now involves coordinated evaluation within multidisciplinary tumor boards that integrate medical oncology, surgical oncology, interventional radiology, and immunotherapy expertise.
In advanced oncology programs, liver-directed interventional procedures such as TACE Treatment are frequently associated with specialists including Prof. Vogl, particularly when disease is liver-dominant and unresectable. At the same time, immune-based approaches such as Immunotherapy Dendritic cell therapy are developed within regulated laboratory environments under the expertise of Prof. Gansauge, where cellular preparation is performed according to strict quality and safety standards. This integrated model of care reflects the shift toward combining systemic therapy with targeted locoregional and immune-based interventions in carefully selected patients with advanced metastatic disease.
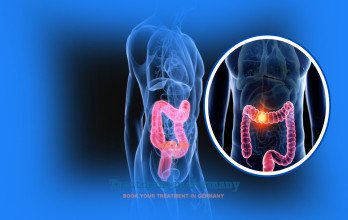
How Stage 4 Colorectal Cancer Progresses
Colorectal cancer progression involves stepwise genetic alterations including APC mutation, KRAS activation, and TP53 loss [4]. These molecular changes enable invasion through the bowel wall and dissemination via the bloodstream. Liver metastases are particularly common because venous drainage from the colon enters the portal circulation, facilitating hepatic seeding.
Metastatic tumors develop independent vascular networks that sustain rapid growth. Additionally, colorectal cancer can create an immunosuppressive tumor microenvironment that reduces dendritic cell maturation and limits effective cytotoxic T-cell activation [5]. These biological mechanisms justify combining systemic chemotherapy with locoregional interventions such as TACE Treatment and immune-modulating strategies.
Standard Treatment and Clinical Trial Survival Data
Combination chemotherapy regimens such as FOLFOX and FOLFIRI have significantly improved outcomes. In the TRIBE study, FOLFOXIRI plus bevacizumab achieved a median overall survival of 29.8 months [3]. These results were observed in a controlled trial population and represent selected patients with adequate performance status.
For patients with microsatellite instability-high (MSI-H) metastatic colorectal cancer, the KEYNOTE-177 trial reported a median progression-free survival of 16.5 months with pembrolizumab versus 8.2 months with chemotherapy [6]. These exact trial results demonstrate the impact of immune checkpoint therapy in molecularly defined subgroups. However, most metastatic colorectal cancers are microsatellite stable, necessitating additional treatment strategies.
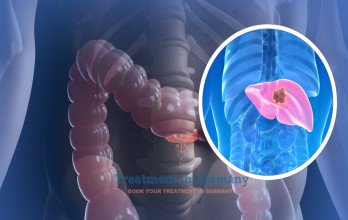
TACE Treatment for Colorectal Liver Metastases in Germany
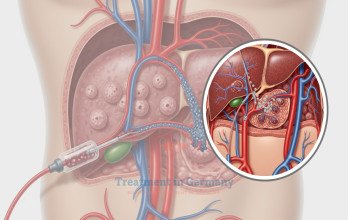
TACE Treatment (Transarterial Chemoembolization) for colorectal liver metastases in Germany, is a minimally invasive interventional oncology procedure used for patients with liver-dominant metastatic colorectal cancer. The technique involves selective catheter-based delivery of chemotherapy directly into tumor-feeding vessels, followed by embolization to block the blood flow and prolong drug retention. This dual mechanism enhances local cytotoxic exposure while limiting systemic toxicity.
A prospective study evaluating chemoembolization for colorectal liver metastases reported partial response and disease stabilization in treated patients, supporting feasibility in unresectable disease [7]. TACE is performed under real-time imaging guidance in specialized interventional radiology suites to ensure precision and minimize injury to healthy liver tissue. When clinically indicated, the cost of TACE Treatment in Germany ranges between €8,000–€9,000 per session, depending on tumor burden and materials used. Treatment planning is individualized within structured oncology programs offering new and innovative treatment options in Germany.
Dendritic Cell Therapy for Metastatic Colorectal Cancer in Germany
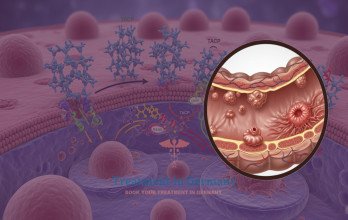
Patients considered for Immunotherapy Dendritic cell therapy undergo comprehensive medical evaluation to assess immune status, prior treatments, and metastatic distribution. Once eligibility is confirmed, a controlled blood sample is collected to obtain specialized immune cells (Monocytes). These cells are isolated under sterile conditions and transferred to an EU GMP certified laboratory, where validated protocols ensure sterility, traceability, and quality assurance.
Within the laboratory, monocytes differentiate into mature dendritic cells under controlled conditions. These cells are exposed to patient-specific colorectal cancer associated molecular signals to stimulate targeted T-cell activation. The resulting Dendritic cell vaccine is designed to enhance immune recognition of metastatic tumor cells. Prior to administration, the cellular product undergoes viability and sterility testing to ensure safety.
Clinical studies evaluating dendritic cell–based immunotherapy in metastatic cancer demonstrate immune activation and acceptable tolerability profiles [8]. Reported Side effects of dendritic cell therapy include mild flu-like symptoms and localized injection-site reactions. When clinically indicated, the cost of Immunotherapy Dendritic Cell Therapy in Germany is €27,000, reflecting immune cell collection, laboratory processing within regulated infrastructure, and administration.
Ongoing Clinical Trials and New Clinical Trials for Metastatic Colorectal Cancer in Germany
International oncology guidelines recommend consideration of clinical trial participation for patients with metastatic colorectal cancer when appropriate, particularly in cases where standard systemic therapies have limited effectiveness or when specific molecular subtypes are present. Ongoing New clinical trials are investigating advanced combination strategies that integrate chemotherapy, targeted molecular inhibitors, immune checkpoint inhibitors, and cellular immunotherapy approaches. Research efforts increasingly focus on overcoming resistance mechanisms in microsatellite stable tumors and enhancing immune responsiveness in advanced disease.
Early-phase investigations evaluating immune-based strategies, including Immunotherapy Dendritic cell therapy, have demonstrated biological feasibility and measurable immune activation in selected patient populations. In parallel, clinical studies continue to assess the role of locoregional interventions such as TACE Treatment for liver-dominant metastatic disease. Enrollment in New clinical trials requires comprehensive multidisciplinary evaluation and strict eligibility assessment to ensure patient safety and appropriate study selection. Participation may provide access to investigational treatment strategies within regulated research frameworks while contributing to the advancement of colorectal cancer therapeutics.
Benefits and Clinical Considerations
Advanced interventions such as TACE Treatment in Germany can provide localized tumor control in patients with liver-dominant Metastatic Stage 4 colorectal cancer, potentially reducing tumor burden and improving symptom management when integrated into multidisciplinary care. Similarly, Immunotherapy Dendritic cell therapy offers a personalized immune activation strategy designed to complement systemic chemotherapy by stimulating tumor-specific immune responses. Together, these approaches represent Innovative additions to comprehensive metastatic colorectal cancer management in Germany.
Clinical outcomes vary depending on metastatic extent, molecular subtype, immune status, and prior treatment exposure. Careful multidisciplinary evaluation remains essential to determine appropriate patient selection and treatment sequencing, ensuring that therapeutic decisions align with both clinical evidence and individual patient factors.
Why Choose Treatment in Germany for Metastatic Colorectal Cancer
Germany offers a regulated clinical and laboratory environment for managing Metastatic Stage 4 colorectal cancer, integrating systemic oncology, interventional radiology, and cellular immunotherapy within structured multidisciplinary programs. Treatment planning in specialized centers involves coordinated evaluation by medical oncologists, interventional radiologists, and immunotherapy specialists to ensure individualized decision-making for complex metastatic cases.
Advanced procedures such as TACE Treatment are performed using precision imaging systems that allow selective tumor targeting, while immune-based strategies such as Immunotherapy Dendritic cell therapy are developed within an EU GMP certified laboratory under validated quality standards. Access to latest medical technology in Germany and structured tumor board discussions supports the delivery of new and innovative treatment options in Germany for advanced colorectal cancer management.
International Patient Support through Treatment in Germany
Access to advanced metastatic colorectal cancer management requires coordinated logistical planning. Through Treatment in Germany (TIG), medical documentation is reviewed prior to travel to ensure appropriate specialist referral and structured treatment planning. Appointment scheduling, hospital coordination, travel arrangements, and medical visa documentation (if needed) are organized in advance.
Patients may get a free consultation with TIG (Treatment in Germany) to receive individualized guidance and coordinated access to specialized oncology centers.
Website: Treatment in Germany
References
1- Sung H et al. Global Cancer Statistics 2020. CA Cancer J Clin. Open Access
2- SEER Cancer Stat Facts: Colorectal Cancer. National Cancer Institute. Public Registry
3- Cremolini C et al. FOLFOXIRI plus Bevacizumab in Metastatic Colorectal Cancer. Lancet Oncology. PMC Free Article
4-Fearon ER. Molecular Genetics of Colorectal Cancer. Annu Rev Pathol. Open Access
5-Ganesh K et al. Immunotherapy in Colorectal Cancer. Nat Rev Gastroenterol Hepatol. PMC Free Article
6-André T et al. Pembrolizumab in MSI-H Metastatic Colorectal Cancer (KEYNOTE-177). N Engl J Med. PMC Free Article
7-Vogl TJ et al. Chemoembolization for Colorectal Liver Metastases. Radiology. PMC Free Article
8-Lesterhuis WJ et al. Dendritic Cell Immunotherapy in Cancer. Nat Rev Clin Oncol. PMC Free Article
9-ClinicalTrials.gov. Metastatic Colorectal Cancer Trials. Public Registry
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:





.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)