Tonsil cancer treatment in Germany including dendritic cell therapy, advanced diagnostics, and coordinated treatment for international patients through Treatment in Germany (TIG).
Tonsil Cancer Treatment: Guide to Advanced Treatment Options in 2026
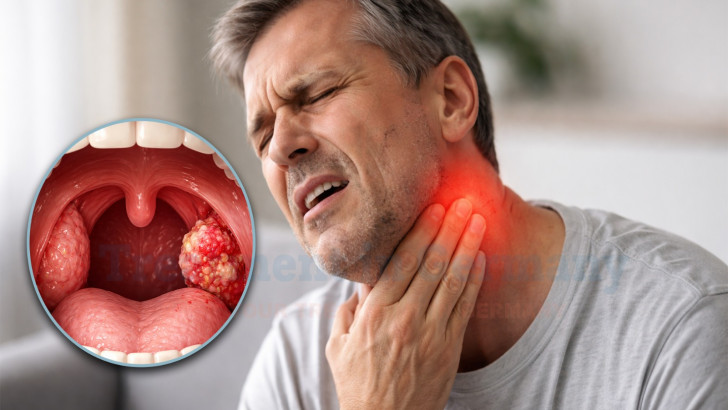
Tonsil cancer is a type of oropharyngeal cancer that develops in the lymphatic tissue located at the back of the throat. The tonsils play a role in immune defense, but like other tissues in the body, their cells can undergo malignant transformation. In recent years, human papillomavirus infection has become an important risk factor alongside smoking and alcohol use. Because the tonsils are part of the throat, early symptoms may involve swallowing difficulty or persistent throat discomfort. Accurate staging and timely intervention are essential to prevent progression and preserve essential functions such as speech and swallowing.
Germany has become a recognized destination for patients seeking new and innovative treatment options for tonsil cancer, particularly for advanced or recurrent disease. Treatment planning is conducted within specialized German healthcare centers and expert-led German Oncology centers, where multidisciplinary tumor boards review each case in detail. Immune-based approaches such as dendritic cell therapy are associated with the clinical expertise of Prof. Gansauge, known for his focused work in advanced cellular immunotherapy. Many international patients travel to Germany for structured treatment pathways, coordinated treatment models, and access to latest medical technology in Germany.
What Is Tonsil Cancer?
Tonsil cancer most commonly arises from squamous cells lining the tonsillar tissue. It may develop due to chronic irritation, tobacco exposure, or viral infection. The disease can remain localized in early stages or extend to nearby lymph nodes in the neck. Early identification significantly improves therapeutic possibilities and long-term control.
Because the tonsils are visible during routine examination, early tumors may be detected during throat evaluation. However, some cases are identified only after lymph node enlargement becomes noticeable. Accurate histological diagnosis is necessary before therapy begins.
Symptoms of Tonsil Cancer
Common early symptoms include persistent sore throat, difficulty swallowing, or a sensation of something stuck in the throat. Some patients experience unilateral throat pain or ear discomfort due to nerve referral patterns. Swelling in the neck caused by lymph node involvement may also be an early sign.
Advanced disease may lead to speech changes, significant swallowing difficulty, or weight loss. Persistent symptoms lasting more than two weeks require structured medical evaluation. Early assessment improves the range of treatment options available.
Advanced Diagnostics for Tonsil Cancer in Germany
Precise staging determines appropriate therapy selection. In Germany, advanced diagnostics include endoscopic examination, biopsy, and imaging such as CT scans, MRI, and PET scans to assess tumor size, lymph node involvement, and distant spread.
These imaging modalities are supported by latest medical technology in Germany, allowing accurate visualization of the oropharyngeal region. Molecular testing may be performed to determine HPV status, which can influence prognosis and treatment decisions. Findings are reviewed collectively within multidisciplinary tumor boards at German oncology centers, ensuring coordinated care planning.
Immunotherapy Dendritic Cell Therapy for Tonsil Cancer in Germany
Immunotherapy has emerged as an evolving strategy in head and neck cancers, including tonsillar malignancies. The dendritic cell vaccine approach is designed to stimulate the immune system to recognize tumor-associated antigens present on tonsil cancer cells. Because certain tumors may suppress immune detection, immune-based strategies aim to enhance systemic immune surveillance and complement standard treatment modalities.
The process begins with the collection of immune cells (monocytes) from the patient’s blood under medical supervision. These monocytes are isolated in a controlled clinical environment to preserve viability and functional integrity. Strict sterile handling ensures cell quality before laboratory processing. Continuous monitoring during collection maintains patient stability and safety.
Collected cells are transported to an EU GMP certified laboratory, where they are cultured under strictly regulated and quality-controlled conditions. Within this environment, the monocytes are exposed to tumor-associated antigens derived from tonsil cancer tissue. This controlled exposure promotes maturation into active dendritic cells capable of presenting cancer markers to T-cells. Comprehensive sterility, purity, and viability testing is performed before clinical use.
Once prepared, the cells are reintroduced into the patient as a personalized dendritic cell vaccine. This therapy is typically integrated alongside surgery, radiation, or systemic therapy rather than replacing them. Continuous monitoring evaluates immune response and overall tolerance. The cost of Dendritic Cell Therapy in Germany is €27,000, covering immune cell collection, laboratory processing, preparation, and administration.
Combining Modern Treatment Strategies in Germany
Tonsil cancer treatment often includes surgery, radiation therapy, or combined chemoradiation depending on stage and tumor characteristics. Early-stage disease may be managed with organ-preserving approaches aimed at maintaining speech and swallowing function. Advanced disease may require multimodal intervention.
Immune-based therapy may be evaluated in selected advanced or recurrent cases as part of a comprehensive management plan. Multidisciplinary coordination within German healthcare centers ensures balanced decision-making between oncologic control and functional preservation. Continuous imaging reassessment supports adaptive treatment planning.
New Clinical Trials for Tonsil Cancer in Germany
Patients with advanced or treatment-resistant disease may be evaluated for participation in new clinical trials for Tonsil cancer in Germany. These structured research programs investigate emerging immunotherapies under strict regulatory oversight.
Clinical research standards align with international frameworks referenced by the American Cancer Society, emphasizing patient safety and scientific transparency. Eligibility requires formal evaluation within accredited German oncology centers. Participation offers access to evolving therapeutic options under controlled conditions.
Limitations and Considerations for Tonsil Cancer Treatment
Not all patients are suitable candidates for immune-based therapies, and careful evaluation is required before initiating advanced treatment. Eligibility depends on tumor stage, lymph node involvement, immune competence, and overall clinical stability. In early-stage tonsil cancer, surgery or radiation therapy alone may provide effective disease control without the need for additional immunotherapy. Decisions are made within multidisciplinary tumor boards at German Oncology centers, where a German specialist evaluates tumor biology, imaging findings, and functional considerations. This structured approach ensures that only patients who may benefit from innovative immune strategies are selected.
Even when considering innovative treatment options for tonsil cancer, therapy must be aligned with realistic clinical goals. Continuous monitoring through imaging such as CT scans, MRI, and PET scans remains essential to evaluate response and detect recurrence early. Management focuses on disease control while preserving essential functions such as speech and swallowing. Treatment planning within German healthcare centers emphasizes balanced decision-making, combining oncologic precision with functional preservation. This individualized strategy is particularly important for international patients seeking advanced but carefully regulated care.
International Patient Support through Treatment in Germany (TIG)
Access to advanced management for tonsil cancer involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as Immunotherapy Dendritic cell therapy. Through Treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This preliminary evaluation by the treating physician helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and Innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:





.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)