Lu177 PSMA therapy in Germany is a targeted nuclear treatment for advanced prostate cancer, delivering radiation directly to PSMA-expressing tumor cells.
What is Lutetium-177 PSMA Therapy?
Lutetium-177 PSMA therapy in Germany radioligand therapy has emerged over the past decade as an important advancement in the management of advanced prostate cancer. It is a targeted radioligand therapy that delivers beta radiation selectively to prostate cancer cells while relatively sparing surrounding healthy tissue. The treatment uses a radioactive isotope called Lutetium-177, conjugated to a ligand that binds to prostate-specific membrane antigen (PSMA) expressed on cancer cells. Once it reaches the cancer cell, it releases radiation that destroys the cell from within. This mechanism distinguishes it from conventional external beam radiotherapy and systemic chemotherapy in terms of targeting precision and toxicity profile.
Understanding how lutetium 177 therapy works in prostate cancer starts with recognizing that a rising PSA level after prostate cancer treatment is often the first signal that the disease is progressing. When a patient experiences a PSA rise despite standard therapies, physicians begin evaluating whether targeted radioligand therapy is the right next step. Germany is considered one of the leading destinations for this treatment due to its early role in developing and implementing PSMA-targeted therapies.
What is PSMA and Why It is Important in Prostate Cancer Treatment
PSMA stands for Prostate-Specific Membrane Antigen, a protein found in very high quantities on the surface of prostate cancer cells. PSMA plays a central role in targeted therapy because it serves as a molecular target that enables selective binding of therapeutic agents to tumor cells. Unlike many other cancers, prostate cancer expresses PSMA at levels far above those seen in normal tissue, making it an ideal target. The more aggressive the cancer, the higher the PSMA expression tends to be, which may enhance the specificity of PSMA-targeted diagnostics and therapy.
PSMA targeted therapy has evolved significantly through years of research and clinical trials progressing from experimental approaches to regulatory-approved clinical applications in several regions. Germany played a leading role in this development, with its research clinics running the first human treatments using PSMA-targeting compounds. Today, patients seeking innovative treatment options in Germany benefit from decades of accumulated clinical experience built over years of research and treatment. This expertise, combined with Germany's strict quality standards, is why so many international patients choose German clinics for this therapy.
Diagnosis and PSMA PET Scan Before Lutetium-177 Therapy
Before therapy begins, patients undergo a comprehensive diagnostic evaluation to confirm eligibility and plan treatment safely. The most important step is the PSMA scan for therapy eligibility, which determines whether the tumor expresses PSMA at sufficient levels for the therapy to work. Only patients with PSMA-positive prostate cancer cells confirmed on imaging are considered suitable for this treatment. The PSMA PET imaging provides highly sensitive whole-body assessment and has been shown to detect metastatic prostate cancer with greater accuracy than conventional imaging modalities [2].
Alongside the PSMA PET scan, patients also require a CT scan and an MRI scan to provide detailed anatomical information about tumor size and location. Laboratory tests are used to assess renal function, hepatic function, and hematologic parameters, which are critical safety indicators. A bone scan may be requested to evaluate skeletal involvement, since bone metastases are common in advanced prostate cancer. In some cases, a biopsy is also performed to confirm the nature of lesions when there is any diagnostic uncertainty. All of these investigations together give the treating team a precise and complete picture before any treatment begins.
Which Stages of Prostate Cancer Can Be Treated with Lutetium-177 PSMA Therapy?
Lutetium-177 (Lu-177) prostate-specific membrane antigen (PSMA) radioligand therapy has robust clinical evidence and is an established treatment option for metastatic prostate cancer .The primary indication is stage 4 prostate cancer that has spread beyond the prostate to lymph nodes, bones, or other organs. More specifically, Lu-177 PSMA radioligand therapy in metastatic castration-resistant prostate cancer (mCRPC) was evaluated in the landmark VISION trial, which demonstrated significant clinical benefit in this patient population [1]. mCRPC refers to cancer that continues to grow despite testosterone being reduced to very low levels through hormone therapy.
Among the most meaningful advanced prostate cancer therapy options in Germany, this therapy now stands as a clinically validated treatment option for patients at this difficult stage. Clinical studies evaluating 177Lu-PSMA therapy have demonstrated improved PSA response rates [1][3], disease control [3], and overall survival [1] in patients with metastatic castration-resistant prostate cancer. Ongoing research is also exploring whether the therapy could be used earlier in the disease course, and German academic centers are actively involved in those trials. Patients treated in Germany may therefore also have access to trial-based therapy when their profile matches current research protocols.
Who is Eligible for Lutetium-177 PSMA Therapy?
The eligibility criteria for lutetium-177 (Lu-177) PSMA radioligand therapy are defined by disease-related and patient-related factors that are carefully assessed prior to treatment initiation. Patient selection in advanced prostate cancer involves a thorough review of treatment history, current imaging, prostate-specific antigen (PSA) kinetics, and overall clinical status. Standard lu-177 treatment candidates are patients with confirmed prostate cancer who have received at least one androgen receptor pathway inhibitor and at least one taxane-based chemotherapy regimen. Their PSMA PET scan must confirm sufficient PSMA expression throughout the disease burden.
Patients also need acceptable kidney function, adequate bone marrow reserves, and a reasonable performance status to tolerate the treatment safely. Those with severely impaired organ function may not be immediately eligible, though treating teams in Germany often work to optimize a patient's condition first. The final decision is always reached through clinical evaluation by the treating physician, who carefully weighs all available data and considers the individual risk-benefit balance. This individualized approach is a defining feature of advanced oncology care at German centers and ensures every patient receives a fair and thorough assessment. These criteria are consistent with patient selection used in major clinical trials of 177Lu-PSMA therapy [1].
If your prostate cancer has progressed after standard treatments, you may be a candidate for PSMA-targeted therapy in Germany.
✔ Get a free medical case review
✔ Reviewed by experienced nuclear medicine specialists
✔ Response within 24–48 hours
How Lutetium-177 PSMA Therapy Works to Target Prostate Cancer Cells
PSMA targeted radioligand therapy works by combining two elements into one molecule: a targeting ligand that recognizes and binds to the PSMA protein, and a radioactive payload that destroys the cell once docked. When injected into the bloodstream, the compound travels throughout the body and latches onto PSMA-expressing cancer cells wherever they are located. The Lutetium-177 then emits beta that penetrate the tumor tissue, inducing DNA damage and ultimately leading to cell death because beta particles have a short tissue range of only a few millimeters, their cytotoxic effect is largely confined to the tumor and adjacent tissue.
This localized delivery is what makes targeted radiation to prostate cancer cells so clinically valuable. A patient with dozens of scattered metastatic sites could not realistically be treated with external beam radiation at every location. Lutetium-177 PSMA therapy, however, reaches all PSMA-expressing sites simultaneously through a single intravenous infusion, using the bloodstream as a delivery network. This makes it uniquely suited to widely metastatic disease with widespread tumor burden and enables simultaneous targeting of multiple PSMA-expressing lesions throughout the body [3].
Scientific Background of Lutetium-177 PSMA Therapy
Lutetium-177 PSMA-617 therapy was primarily developed at the German Cancer Research Center (DKFZ) and University Hospital Heidelberg in Germany, where early studies demonstrated its ability to selectively target PSMA-expressing metastatic prostate cancer cells. Early clinical studies demonstrated promising results, which contributed to broader international interest and subsequent large-scale clinical trials
Based on strong clinical evidence, in March 2022, 177Lu-PSMA-617 (lutetium Lu 177 vipivotide tetraxetan) was approved by the U.S. Food and Drug Administration (FDA) for the treatment of patients with metastatic castration-resistant prostate cancer who had previously received androgen receptor pathway inhibition and taxane-based chemotherapy. This approval marked a major milestone in targeted cancer treatment, making this therapy widely accessible and reinforcing Germany’s leading role in its development and clinical application.
Lutetium-177 PSMA Therapy vs Chemotherapy
When comparing Lu- 177 PSMA radioligand therapy with chemotherapy in prostate cancer, there are significant differences in mechanism of action and treatment tolerability. Chemotherapy agents such as docetaxel and cabazitaxel act on rapidly dividing cells systemically, which contributes to adverse effects such as alopecia, nausea, and bone marrow suppression. For patients already weakened by advanced disease, tolerating multiple chemotherapy cycles is genuinely difficult. As an alternative to chemotherapy, Lu-177 PSMA radioligand therapy offers a more targeted mechanism of action and is generally associated with a different and often more favorable toxicity profile.
In the therapy trial, Lu-177 PSMA-617 was compared with cabazitaxel in patients with metastatic castration-resistant prostate cancer, demonstrating higher PSA response rates and fewer grade 3–4 adverse events in the radioligand therapy group.[5]. This positions Lutetium-177 PSMA therapy firmly as a targeted therapy vs conventional cancer treatment that delivers both clinical benefit and a better treatment experience in appropriately selected patients. Rather than replacing chemotherapy entirely, it offers an additional active treatment option with a completely different mechanism of action, giving patients and physicians more tools to manage the disease over time.
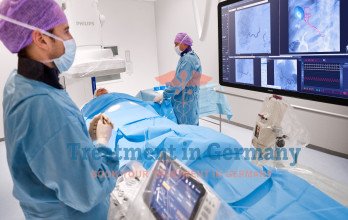
Step-by-Step Lutetium-177 PSMA Therapy Procedure and Treatment Cycles
The lutetium-177 (Lu-177) PSMA radioligand therapy procedure begins once pre- treatment evaluation confirms eligibility. The lu-177 treatment cycles are administered every 6 to 8 weeks, allowing time for radiation to act and the body to recover before the next dose. Most patients receive up to six cycles in total, though this is adjusted based on response and tolerability at each assessment point. The interval between cycles also serves as a monitoring window, during which PSA levels and organ function are checked to ensure treatment is proceeding safely.
Each cycle involves the intravenous administration of lutetium 177 therapy through a peripheral vein over approximately 30 minutes, carried out in a shielded nuclear medicine suite. The outpatient prostate cancer treatment format entails many patients remaining under observation for one to two nights before discharge. The treatment process of lutetium 177 PSMA therapy in Germany is organized and patient-centered, with dedicated case managers supporting international patients throughout. Post-infusion dosimetry scans are often performed to confirm proper distribution of the compound and to guide dosing adjustments in subsequent cycles.
Can Lutetium-177 PSMA Therapy Be Combined with Other Treatments?
Lutetium 177 combination therapy for prostate cancer is an active area of clinicalresearch, with several promising strategies currently under investigation. In clinical practice, Lu-177 PSMA therapy is typically administered alongside ongoing androgen deprivation therapy (ADT), as castrate testosterone levels help suppress tumor proliferation. Combination approaches involving Lu-177 PSMA therapy and chemotherapy are being investigated in clinical trials, including hypotheses that some chemotherapeutic agents may modulate PSMA expression and tumor biology, potentially influencing radioligand uptake.
Multimodal prostate cancer treatment strategies combining Lutetium-177 with PARP inhibitors are another area of active interest, since PARP inhibitors impair DNA repair and may amplify the radiation effect. Integrated oncology treatment in Germany is delivered through multidisciplinary tumor boards that review each patient individually and ensure combination strategies are evidence-based. German centers are participating in several international trials investigating these combinations, meaning patients treated there may benefit from access to experimental approaches not yet widely available elsewhere.
Is It Necessary to Repeat Lutetium-177 PSMA Therapy?
The need for repeating treatment depends on how well the patient responds and tolerates the therapy. Most patients receive multiple cycles of Lu-177 PSMA radioligand therapy, depending on tumor burden and disease stage, with regular assessments between cycles to monitor safety and treatment response. Some may require fewer cycles, while others with more advanced disease may benefit from the full course.
The treatment plan is always adjusted to achieve the best possible disease control while minimizing side effects. If the disease progresses later and PSMA expression is still present, additional treatment may be considered. All decisions are made in close consultation between the patient and the medical team to ensure a individualized treatment approach.
Side Effects and Safety of Lutetium-177 PSMA Therapy
The most commonly reported lutetium 177 PSMA side effects are fatigue, dry mouth, nausea, and mild hematologic toxicity such as anemia and thrombocytopenia. These prostate cancer treatment side effects are generally manageable and typically improve between treatment cycles. Dry mouth is a characteristic side effect because PSMA is expressed in salivary glands as well as cancer cells, leading to off-target radiolig and uptake and radiation exposure of salivary tissue. Patients are advised to stay well hydrated and report any worsening symptoms promptly so adjustments can be made.
More serious lutetium 177 PSMA risks and complications include kidney toxicity and bone marrow suppression, particularly in patients with pre-existing impairment in these areas. Renal function is therefore closely monitored before each cycle, and therapy may be adjusted or temporarily withheld if significant deterioration in renal parameters occurs German centers manage these risks through rigorous safety protocols, and their experience with large patient volumes means that side effects are identified and addressed quickly. Clinical dosimetry and safety studies have confirmed that 177Lu-PSMA therapy has demonstrated a favorable safety profile with manageable toxicity in clinical studies [4].
What to Expect After Lutetium-177 PSMA Therapy and Recovery
Recovery after Lu-177 PSMA radioligand therapy is generally well tolerated, with most patients experiencing mild, transient adverse effects such as fatigue or nausea for a few days. Many are able to return to normal daily activities shortly after each cycle. Patients receiving Lu-177 patient recovery in Germany benefit from structured support, including guidance on managing side effects and access to their care team if needed.
Post therapy monitoring in prostate cancer is an important part of recovery, involving regular PSA tests and a follow-up PSMA PET scan to assess response. Many patients also notice improvement in symptoms such as bone pain, leading to better daily comfort and quality of life.
Cost of Lutetium-177 PSMA Therapy in Germany
The cost of Lutetium-177 PSMA therapy in Germany is approximately €19,000, depending on the number of cycles and overall treatment plan. This typically includes the radiopharmaceutical, advanced nuclear medicine facilities, and expert medical supervision under experienced specialists such as Prof. Stefan Dresel, a highly regarded expert in nuclear medicine with extensive experience in PSMA-based therapies.
Additional costs such as diagnostics, travel and coordination services may vary based on individual needsLutetium-177 PSMA Therapy. Patients are usually provided with a detailed estimate in advance to ensure transparency and proper planning before starting treatment.
Success Rate and Clinical Outcomes of Lutetium-177 PSMA Therapy
The lutetium 177 PSMA success rate is supported by robust data from large clinical trials. The VISION trial showed that 177Lu-PSMA-617 improved radiographic progression-free survival (median 8.7 months versus 3.4 months in controls) and overall survival (median 15.3 months versus 11.3 months) compared to standard care alone [1]. PSA reduction rates with lu-177 therapy are also meaningful, with more than 46% of treated patients in the VISION trial achieving a PSA decline of 50% or greater from baseline [1]. These are clinically significant results however, individual outcomes vary depending on disease burden, prior therapies, and patient characteristics.
The clinical outcomes of prostate cancer in Germany observed in real-world practice at major German hospitals have been broadly consistent with these trial results. For international patients weighing their options, this published data from both trials and real-world settings provides genuine confidence. Treatment outcomes in advanced prostate cancer depend heavily on PSMA expression levels, prior treatment history, and overall health, which is why thorough pre-treatment assessment is so important to selecting the patients most likely to benefit from this therapy.
How to know if Lutetium-177 PSMA Therapy is working
PSA response after lutetium 177 therapy is the most accessible early indicator of treatment benefit and is measured regularly throughout the treatment course. A sustained decline in prostate-specific antigen (PSA) of ≥50% from baseline is generally considered a clinically meaningful biochemical response. However, PSA behavior over time must be read carefully, because a temporary PSA flare can occur after the first cycle before the value subsequently falls. This flare does not necessarily indicate treatment failure and should not, in isolation, prompt premature discontinuation of therapy.
PSMA PET scan follow-up performed around three months after completing the course provides the most comprehensive imaging-based response assessment, showing directly how metastatic sites have responded. Prostate cancer treatment monitoring combines these imaging findings with PSA trends, symptom assessments, and blood test results to build a full picture of lu-177 therapy effectiveness. The treating team reviews all of this information with the patient clearly and uses it to guide decisions about additional treatment, ongoing surveillance, or a change in strategy if the response has been insufficient.
Best Hospitals in Germany for Lutetium-177 PSMA Therapy
Germany is home to highly advanced hospitals for Lutetium-177 PSMA therapy, offering world-class care for patients with advanced prostate cancer. These specialized centers are equipped with modern nuclear medicine facilities, including dedicated treatment units and advanced imaging systems. Multidisciplinary teams of experts work together to design personalized treatment plans based on each patient’s condition.
The strength of prostate cancer treatment lies in its strict quality standards and well-structured healthcare system. Certified cancer centers follow rigorous protocols for safety, treatment outcomes, and continuous improvement. This ensures that international patients receive reliable, transparent, and high-quality care supported by ongoing clinical research and expertise in advanced oncology in Germany.
Why Patients Choose Germany for Lutetium-177 PSMA Therapy
Germany is widely recognized for its structured healthcare system, advanced medical infrastructure, and strict regulatory standards, making it a trusted destination for patients seeking prostate cancer treatment in Germany. The country has played a key role in the development of Lutetium-177 PSMA therapy, supported by experienced specialists and modern nuclear medicine facilities. This ensures that treatment is delivered with precision, safety, and consistency under well-regulated clinical conditions. Patients from countries such as the UK and USA often choose Germany for access to timely and advanced therapies that may not be readily available in their home healthcare systems.
To make the process seamless for international patients, dedicated coordination services are available throughout the treatment journey. TIG GmbH help connect patients with certified hospitals and experienced specialists across Germany. With a wide network of clinics, these services offer transparent planning, personalized support, and assistance with medical evaluation, travel, and follow-up care. This structured and patient-focused approach ensures that individuals receive high-quality treatment in a smooth and well-organized manner within the landscape of advanced oncology in Germany.
Why Choose Us & Your Treatment Journey
Choosing the right medical coordination partner is essential when planning treatment abroad. We focus on providing a structured, transparent, and patient-centered experience for international patients, ensuring every step is organized, clear, and stress-free. Our goal is to simplify the entire process while maintaining high standards of care, communication, and reliability.
Quick Service Performance: Fast and efficient coordination
Fast Appointment Arrangements: Timely scheduling with specialists
Customized Medical Program: Personalized treatment planning
Visa Support (if needed): Assistance with travel documentation
Direct Hospital Payments: Secure and transparent payment process
Choice of Doctors: Access to experienced specialists
Transparent Invoicing: No hidden costs, full clarity
Multilingual Support: Smooth communication throughout
Follow-Up Support: Continued care after treatment
Dedicated Coordinator: Personal assistance at every step
Your journey with us begins by sharing your medical reports, followed by expert review and treatment planning. We then manage hospital coordination, travel, accommodation, and appointments to ensure a seamless experience. During your stay, our team supports you throughout consultations and procedures, and after treatment, we assist with reports, follow-up care, and ongoing guidance to ensure continuity and peace of mind.
Start Your Treatment Journey in Germany
From medical review to travel and hospital coordination, we handle everything.
✔ Fast-track appointments
✔ Visa & travel support
✔ Dedicated case manager
References
Useful Links
Lutetium-177 PSMA Therapy in Germany
Lutetium-177 PSMA Therapy for Prostate Cancer in Germany
Prostate Cancer treatment with Lutetium Lu-177 PSMA therapy in Germany
Best Innovative Treatments for Prostate Cancer in Early Stages
Comprehensive Guide to Stage 4 Prostate Cancer: Treatment Options
Patients may also request a free consultation with TIG (Treatment in Germany) to receive individualized guidance and coordinated access to treatment in Germany.










.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)