Explore clinical trials for prostate cancer treatment in Germany, including dendritic cell immunotherapy, TACP treatment, PSMA therapies, and complete logistical support for international patients through TIG.
Clinical Trials for Prostate Cancer Treatment in Germany
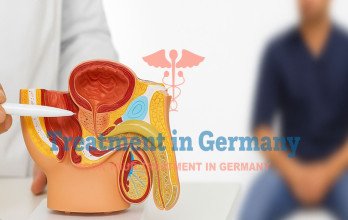
Prostate cancer is one of the most commonly diagnosed cancers in men, and in advanced stages it requires carefully planned, long-term treatment strategies. When the disease progresses beyond the prostate gland, standard therapies may no longer provide sufficient disease control, which is why clinical trials become an important consideration.
Germany has established itself as a leading destination for clinical trials for prostate cancer treatment, supported by structured diagnostics, regulated research protocols, and access to innovative treatment options in Germany. Clinical trial programs in Germany are guided by experienced specialist teams working within German oncology centers and advanced German healthcare centers, using the latest medical technology in Germany.
Specialist-led programs include immune-based therapies and interventional oncology approaches, overseen by experts such as Prof. Gansauge, known for advanced cell-based cancer immunotherapy, Prof. Vogl, widely recognized for interventional oncology procedures, and Prof. Dr. Stefan Dresel, associated with PSMA-based radioligand therapies. International patients benefit from coordinated access and complete logistical support through Treatment in Germany (TIG) at www.treatmentingermany.de , ensuring structured care without administrative complexity.
Understanding Clinical Trials for Prostate Cancer
Clinical trials evaluate new or optimized treatment approaches under controlled medical supervision. In prostate cancer, trials may focus on improving disease control, targeting cancer-specific markers, or enhancing immune recognition. Participation is based on strict eligibility criteria, including disease stage, overall health, diagnostic findings, and previous therapy history.
Germany is often selected for prostate cancer trials because of its transparent research framework and strong emphasis on patient safety. Diagnostic evaluation typically includes Diagnostics, supported by CT scans, MRI, PET scans, allowing specialists to assess disease activity and suitability for trial enrollment. These structured pathways provide clarity for patients seeking stage 4 prostate cancer new treatments in Germany.
Why International Patients Choose Germany for Prostate Cancer Trials
International patients choose Germany because of its regulated research environment, access to best oncologist in Germany networks, and treatment delivered through specialized German oncology centers. Transparent protocols and structured follow-up provide confidence for patients navigating advanced disease.
Complete logistical coordination, including medical travel planning and documentation, is managed through Treatment in Germany (TIG), allowing patients to focus on treatment rather than administrative tasks.
Immunotherapy Dendritic Cell Therapy in Prostate Cancer Clinical Trials
Immunotherapy Dendritic Cell Therapy in Germany is evaluated in prostate cancer clinical trials as a personalized immune-based approach. Rather than directly attacking tumors, this therapy supports the immune system’s ability to recognize prostate cancer–related markers and maintain immune surveillance over time.
The process begins with a medical review to confirm suitability. A blood sample is collected, and immune cells are isolated and processed in an EU GMP certified laboratory. These cells are trained using prostate cancer–specific antigens so they can present cancer signals more effectively to immune cells. After safety and quality checks, the prepared cells are administered back to the patient as a personalized dendritic cell vaccine under medical supervision.
In clinical trials, immune response and disease behavior are monitored through structured follow-up. Outside trial settings, the cost of Immunotherapy Dendritic Cell Therapy in Germany ranges between approximately €24,000 and €26,000, including immune cell collection, laboratory processing, vaccine preparation, and administration. Reported side effects of dendritic cell therapy are generally mild and may include temporary fatigue or localized reactions.
TACP Treatment for Prostate Cancer in Germany
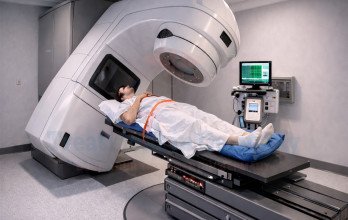
TACP Treatment (Transarterial Chemoperfusion) is evaluated in prostate cancer research as a regional treatment approach designed to deliver chemotherapy directly to tumor-related circulation. This technique aims to increase local drug concentration while reducing unnecessary systemic exposure.
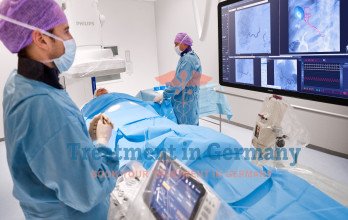
Before treatment, specialists assess diagnostic imaging and overall health to determine whether localized tumor control may support disease management. During the procedure, a catheter is inserted through a small access point and guided under imaging control to the artery supplying the tumor region. Chemotherapy is then infused in a controlled manner while maintaining blood flow to surrounding tissue.
Patients are monitored during and after the procedure, and follow-up imaging helps determine treatment response. Outside trials, the cost of TACP Treatment in Germany typically ranges between €8,000 and €9,000 per session, depending on treatment planning and complexity.
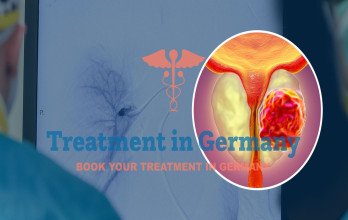
PSMA-Based Radioligand Therapy Trials in Germany
Lutetium-177 PSMA Therapy in Clinical Trials
Lutetium-177 PSMA Therapy is studied in prostate cancer clinical trials for patients whose tumors express PSMA markers. The therapy delivers targeted radiation to cancer cells while limiting exposure to healthy tissue. It is evaluated using structured protocols to assess safety and disease response.
In non-trial settings, the cost of Lutetium-177 PSMA Therapy in Germany is approximately €19,000 per session. This approach is often evaluated when prostate cancer shows biochemical progression, including rising PSA levels after prostate cancer diagnosis.
Actinium-225 PSMA Therapy in Clinical Research
Actinium-225 PSMA Therapy is an advanced targeted alpha therapy evaluated in selected prostate cancer cases. It delivers highly localized radiation to PSMA-expressing cancer cells under strict medical supervision. German centers apply careful eligibility assessment due to the therapy’s potency.
This approach represents one of the most advanced innovative treatment options in Germany for metastatic prostate cancer and is considered within structured research environments.
Role of Diagnostics in Prostate Cancer Clinical Trials
Accurate diagnostics are essential for trial eligibility and treatment planning. German centers rely on integrated Diagnostics, including CT scans, MRI, PET scans, to evaluate disease distribution, tumor activity, and response monitoring. This structured diagnostic approach supports safer trial participation and clearer treatment decisions.
Combining Advanced Treatments for Stage 4 Prostate Cancer in Germany
In some cases, doctors may consider combining immune-based therapy with interventional or nuclear medicine approaches. Combination strategies are evaluated cautiously and tailored to the individual patient’s disease pattern, tolerance, and treatment history. This integrated planning reflects Germany’s emphasis on structured and personalized oncology treatment.
Support for International Patients through Treatment in Germany
For international patients, managing cancer treatment abroad can be overwhelming. TIG, Treatment in Germany at www.treatmentingermany.de , provides complete logistical coordination, including hospital scheduling, travel planning, and medical visa assistance if required. This allows patients to focus on treatment decisions rather than administrative challenges.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:










.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)