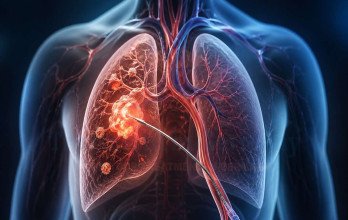
Transpulmonary Chemoembolization (TPCE) offers a targeted option to control lung tumors when standard treatments are no longer effective.
What is Transpulmonary Chemoembolization (TPCE)?
When surgery or systemic treatments are no longer effective, targeted options like Transpulmonary Chemoembolization (TPCE) can help control lung tumors and metastases. It is a specialized interventional oncology procedure designed to deliver chemotherapy directly into the blood vessels supplying lung tumors, followed by controlled embolization to block tumor blood flow. This approach is particularly considered in Germany, where advanced interventional techniques are integrated into cancer care for patients who are not suitable for surgery or whose disease has progressed despite systemic therapies. By concentrating chemotherapy within the tumor, TPCE allows higher local drug exposure while reducing systemic toxicity, which is a key concern in advanced cancer treatment.
Clinical evaluation provides realistic insight into its role. A study assessing repeated TPCE procedures reported that 55% of patients demonstrated partial tumor response based on RECIST criteria, indicating measurable tumor reduction rather than complete remission [1]. This positions TPCE as a form of chemoembolization for lung cancer and a targeted treatment, particularly useful in the treatment of lung metastases, where the clinical goal is to control disease progression and improve patient stability rather than promise cure.
Get a Free TPCE Eligibility Review
If you have been diagnosed with lung cancer or lung metastases and want to know whether TPCE is suitable for your case, you can send your medical reports for a specialist review. You will receive a personalized assessment, treatment options, and next steps.
How TPCE Targets Lung Tumors and Pulmonary Metastatic Disease
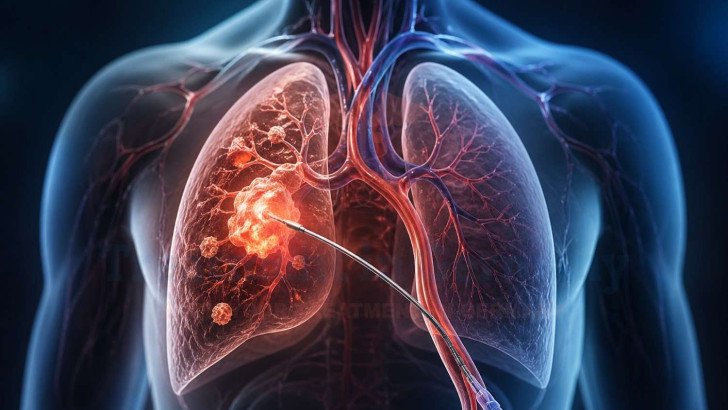
The potential effectiveness of transpulmonary chemoembolization (TPCE) is related to its ability to target tumor-feeding vasculature, making it relevant in selected lung tumors and pulmonary metastases. During the TPCE procedure, a catheter is inserted into the vascular system and guided under imaging toward tumor-feeding pulmonary vessels. Chemotherapy is then delivered directly into this region, ensuring a high concentration of the drug within the tumor environment while minimizing exposure to healthy tissues.
Following this, embolization is performed to block blood flow, which enhances drug retention and limits tumor growth. A technical study describing this method confirms improved intratumoral drug exposure and localized tumor control through combined chemoembolization techniques [2]. This supports its role as a targeted treatment for lung metastases, especially in complex cases such as advanced or recurrent non-small cell lung cancer (NSCLC) treatment in Germany and small cell lung cancer (SCLC) treatment in Germany, where achieving localized disease control is clinically significant.
Why TPCE is Emerging as an Important Option in Interventional Oncology
TPCE is increasingly being incorporated into targeted cancer therapy for lung cancer, particularly for patients with limited treatment options. One of the key benefits of TPCE procedure is its ability to deliver therapy directly to tumor sites while minimizing systemic side effects, which is especially relevant for patients with unresectable lung metastases. This localized approach allows repeated treatment sessions without the cumulative toxicity often associated with systemic chemotherapy.
It is important to interpret outcomes carefully. Clinical findings describe TPCE effectiveness in terms of tumor shrinkage or stabilization rather than complete elimination. Observations such as rapid shrinkage in tumor size refer to measurable reduction, while the ability to stop tumor progression reflects disease stabilization over time [1]. For patients, this means TPCE is best understood as a method to manage cancer progression within a broader treatment strategy.
TPCE Compared to Systemic Chemotherapy in Lung and Metastatic Tumors
When comparing transpulmonary chemoembolization (TPCE) with systemic chemotherapy, key differences relate to treatment distribution and toxicity profiles. Systemic chemotherapy circulates throughout the body, affecting both cancerous and healthy cells, which can result in widespread side effects. In contrast, TPCE focuses treatment directly at the tumor site, allowing a more localized and controlled therapeutic effect.
According to established oncology guidelines, systemic therapy remains the primary treatment for widespread disease, while localized approaches are used to control specific tumor sites [4], TPCE may be considered as a locoregional option, particularly when disease burden is predominantly confined to the lungs. It is typically used alongside other therapies rather than as a standalone replacement.
Which Types of Lung Tumors and Metastatic Cancers Can Be Treated with TPCE?
TPCE is used in a wide range of pulmonary malignancies, particularlymetastases originating from primary tumors, including colorectal carcinoma, breast cancer, renal cellular carcinoma, thyroid cancer, cholangiocellular carcinoma and others. These cancers frequently spread to the lungs and may not be suitable for surgical removal due to the number or distribution of lesions.
The procedure is also considered in selected cases of primary lung tumors where conventional treatment options are limited. Clinical studies evaluating TPCE include patients with diverse tumor origins, and outcomes are assessed based on imaging response rather than tumor-specific cure rates [1]. This highlights that TPCE is primarily used as a disease control strategy in complex and advanced cancer settings.
Useful Links
- Treatment Options for Lung Cancer in Germany
- Lung Metastases from Breast Cancer: A Complete Treatment Guide 2026
- Treatments for Stage 4 Non–Small Cell Lung Cancer
- What are the new Treatment Options for Stage 4 Lung Cancer?
Diagnosis and Pre-Treatment Evaluation before TPCE Procedure
Before undergoing TPCE, patients require a detailed assessment that includes lung cancer diagnosis tests, such as MRI Scan, CT scan, pathological biopsy, blood work. CT imaging plays a central role in identifying tumor location, size, and vascular supply, which directly influences procedural planning.
Biopsy is used to confirm the tumor type and guide treatment decisions, while blood work ensures that organ function is adequate for the procedure. This structured evaluation is essential to determine whether the patient is a suitable candidate for TPCE and to minimize potential risks. Careful pre-treatment planning is a key factor in achieving safe and effective outcomes.
Step-by-Step Treatment Process and Duration of TPCE Treatment
TPCE follows a structured interventional process, particularly in cases involving lung metastases from various primary cancers. The procedure begins with catheter insertion into a blood vessel, followed by navigation to the tumor site using imaging guidance. Once the catheter is correctly positioned, Chemotherapy is delivered into the arterial supply feeding the tumor, ensuring precise targeting.
After drug delivery, embolization is performed to block the blood flow and enhance treatment effectiveness. The procedure typically takes a few hours depending on tumor complexity. Patients are monitored after treatment, and follow-up imaging is scheduled to evaluate response. This systemic approach ensures accurate therapy delivery and consistent outcome assessment.
Can TPCE Be Used Alongside Other Cancer Therapies?
TPCE is often integrated into combined treatment strategies, including TPCE combined with chemotherapy or immunotherapy, particularly in advanced stage cancers. This allows multiple therapies to work together, targeting cancer through different mechanisms.
This approach aligns with multimodal cancer treatment, where localized and systemic therapies are combined based on individual patient needs. Clinical literature supports this integrated strategy in improving disease control, especially in complex cancer cases [3].
Who is Eligible for TPCE Treatment?
Determining the right candidate for TPCE therapy requires a careful and individualized approach based on tumor characteristics and overall patient condition. A detailed clinical evaluation by the treating physician is essential to assess whether the procedure is both safe and potentially beneficial. TPCE is generally considered for patients who are not candidates for surgery or whose disease has not responded adequately to systemic treatments.
It is most often used in advanced stage cancer or intermediate-stage liver cancer or selected advanced cases, particularly when tumor involvement is predominantly within the lungs. The TPCE eligibility criteria typically include stable organ function, controlled comorbid conditions, and the ability to tolerate a minimally invasive procedure. For patients, this section is crucial because it directly answers whether TPCE may be a realistic option, helping move from general information toward a treatment decision.
Not sure if you qualify for TPCE?
Eligibility depends on your tumor type, stage, and overall condition. Our specialists can review your case and guide you on whether this treatment is appropriate.
Request your case evaluation today.
How to know if TPCE Treatment is Working?
Evaluating treatment success requires objective follow-up rather than relying on symptoms alone. In clinical practice, TPCE results are assessed using imaging techniques by use of contrast-enhanced imaging (CT or MRI) assessing viable tumor (enhancement) these scans are usually performed several weeks following the procedure to allow for an accurate evaluation of response.
The concept of tumor response after TPCE treatment includes partial response, stable disease, or progression based on standardized imaging criteria. Studies examining TPCE effectiveness in lung cancer have shown that tumor reduction or stabilization is commonly observed, rather than complete disappearance [1]. This structured evaluation ensures that further treatment decisions are based on measurable outcomes rather than assumptions.
Clinical Outcomes and Success Rate of TPCE Treatment
The TPCE success rate is best understood in terms of disease control rather than cure. Clinical data reporting the success rate of TPCE therapy in Germany indicate that a proportion of patients experience partial tumor response or stabilization following repeated treatment sessions [1]. These outcomes are measured through imaging and reflect changes in tumor size over time.
Reports involving patients from USA and other international countries suggest that TPCE can help maintain disease stability in selected cases. The survival outlook of TPCE depends on multiple clinical factors, including tumor type, extent of disease, and overall patient condition. Ongoing research, including new studies for stage 4 lung cancer in Germany, continues to explore how TPCE fits into combined treatment strategies for advanced disease.
Side Effects and Safety of (TPCE) Treatment
The chemoembolization risks associated with TPCE are generally considered manageable when the procedure is performed in experienced medical clinics in Germany. The most common adverse effects include fatigue, low-grade fever, nausea, and abdominal discomfort, collectively known as post-embolization syndrome. These effects are related to the localized nature of the treatment and typically resolve with supportive care.
The TPCE side effects profile differs from systemic chemotherapy because the treatment is concentrated in the tumor region rather than distributed throughout the body. Clinical studies have reported that adverse events are generally controlled and consistent with interventional procedures when appropriate protocols are followed [2]. This highlights the importance of undergoing treatment in specialized centers with expertise in interventional oncology.
Cost of TPCE Treatment in Germany
The cost of TPCE therapyranges between €8,000 and €9,000 per session in Germany, which includes imaging guidance, catheter-based intervention, chemotherapy delivery, embolization materials, and post-procedure monitoring.
The frequency of treatment varies depending on tumor response, which is why how often TPCE is repeated differs from patient to patient. In clinical practice, procedures are often performed by experienced specialists such as Prof. Vogl, known for expertise in embolization therapies. Patients are encouraged to review detailed treatment plans to understand the expected number of sessions and overall cost structure.
Scientific Background of TPCE and How It Targets Tumor Blood Supply
TPCE originated in the early 2000s in Germany, where advancements in interventional oncology enabled targeted drug delivery through catheter-based techniques. At University Hospital Frankfurt, Prof. Vogl played a key role in advancing this technique, particularly for patients with lung metastases and primary lung tumors that could not be surgically removed.
The scientific principle behind TPCE involves delivering chemotherapy directly into tumor-feeding vessels while simultaneously blocking blood flow to enhance drug retention. This dual mechanism increases local drug concentration and limits systemic toxicity. Clinical and technical studies have described this targeted approach as an effective method for controlling tumor growth in selected patients [2].
Is It Necessary to Repeat TPCE Treatment?
In many cases, TPCE is performed as part of a treatment strategy involving multiple TPCE sessions. The decision to repeat treatment is based on follow-up imaging, tumor response, and overall clinical condition. Repeat procedures are often recommended when residual tumor activity or new lesions are identified.
The need for additional sessions varies depending on tumor burden and cancer stage, as well as patient-specific factors. This flexible approach allows physicians to adapt treatment over time, aiming to maintain disease control while minimizing risk. Repeat TPCE is therefore considered part of a long-term management strategy rather than a one-time intervention.
Why Patients Choose Germany for TPCE Treatment and Our Support Services
Germany is known for advanced and structured cancer treatment, especially in interventional oncology procedures like TPCE. Patients benefit from experienced specialists, modern imaging technology, and standardized treatment protocols that ensure safety and precision. High-volume centers further enhance treatment accuracy, making Germany a trusted option for managing complex cancer cases.
To make the process easier for international patients, TIG provide complete support at every step of the journey:
- Fast appointment scheduling with experienced specialists
- Customized treatment plans based on your condition
- Visa support (if required)
- Direct hospital payments with transparent billing
- Choice of preferred doctors
- Continuous follow-up support
- Multilingual assistance for clear communication
Start Your TPCE Treatment Journey
Getting started is simple:
Share your medical reports
Receive expert evaluation
Get a personalized treatment plan
Our team will assist you with appointments, travel coordination, and complete treatment support in Germany.
Request your free consultation today.
References (Properly Formatted with DOI)
National Cancer Institute. Non-small cell lung cancer treatment (PDQ®). 2023. Available from: https://www.cancer.gov
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:








.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)