Explore treatments options for stage 4 non–small cell lung cancer in Germany, including TPCE and TACP therapies through TIG for International Patients.
Treatments for Stage 4 Non–Small Cell Lung Cancer
Stage 4 metastatic non small cell lung cancer represents the most advanced stage of non–small cell lung cancer (NSCLC) and is defined by the spread of cancer cells beyond the lung to distant organs such as the brain, bones, liver, adrenal glands, or the opposite lung. At this stage, the disease is systemic and requires coordinated, multidisciplinary medical management. According to SEER population-based survival data, the overall 5-year relative survival rate for lung and bronchus cancer is 26.4%, while the 5-year relative survival rate for Stage IV non–small cell lung cancer is 5.8% [1]. These statistics demonstrate the marked difference in outcomes between localized and metastatic disease and highlight the importance of comprehensive treatment planning.
Advanced NSCLC is frequently diagnosed at late stages. In a real-world cohort study of advanced NSCLC patients, 76.0% were diagnosed at Stage IV [2]. This emphasizes the need for structured treatment pathways once metastatic disease is identified. Within Germany’s healthcare system, lung cancer care is delivered through multidisciplinary tumor boards integrating medical oncology, interventional radiology, pathology, thoracic surgery, and immunotherapy specialists. Interventional oncology procedures are associated with experts such as Prof. Vogl, while cellular immunotherapy programs have been developed in association with Prof. Gansauge, both operating within regulated institutional environments.
Understanding Stage 4 NSCLC
Stage 4 metastatic non small cell lung cancer occurs when malignant lung epithelial cells invade local tissues and enter vascular or lymphatic circulation, establishing metastatic deposits in distant organs. Because tumor spread is systemic, treatment strategies must address both visible lesions and microscopic disease.
The symptoms of stage 4 non–small cell lung cancer vary according to metastatic involvement. Common respiratory symptoms include persistent cough, dyspnea, and chest discomfort. Bone metastases often cause localized pain or fractures, while brain metastases may result in headaches or neurological changes. General systemic manifestations such as fatigue and unintended weight loss are also frequent. Diagnosis relies on imaging, histopathological confirmation, and molecular profiling to guide individualized therapy.
Standard Treatment Options in Germany
Standard treatment options for stage 4 non–small cell lung cancer include systemic chemotherapy, targeted therapy for tumors with actionable mutations, and immune checkpoint inhibitors for biomarker-selected patients. Because metastatic NSCLC is systemic, pharmacologic therapy remains foundational.
Radiotherapy may be applied to control symptoms arising from metastatic lesions. Treatment decisions depend on molecular characteristics, overall performance status, organ function, and previous therapies received. Within Germany’s healthcare system, therapy planning occurs in multidisciplinary settings to ensure safety, coordination, and regulatory oversight.
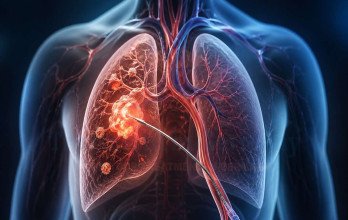
TPCE Treatment (Transpulmonary Chemoembolization) for non small cell lung cancer in Germany
TPCE Treatment (Transpulmonary Chemoembolization) for non small cell lung cancer in Germany is a catheter-based interventional oncology procedure designed to increase local chemotherapy concentration while reducing systemic exposure. The intervention is performed in specialized angiography units equipped with latest medical technology in Germany, allowing detailed vascular mapping and precise catheter placement.
The procedure begins with vascular access under sterile conditions. A catheter is advanced into the pulmonary arterial circulation under fluoroscopic guidance. Selective angiography identifies tumor-feeding branches, and a microcatheter is positioned directly within these vessels. Chemotherapeutic agents are infused into the tumor-supplying arteries to maximize intratumoral exposure. Following chemotherapy infusion, embolic particles are administered to partially reduce blood flow to the tumor. This prolongs drug retention and induces ischemic stress within malignant tissue. Continuous cardiopulmonary monitoring ensures procedural safety, and post-intervention imaging evaluates treatment response.
The cost of TPCE Treatment in Germany typically ranges between €8,000 and €9,000 per session, depending on individual treatment planning and disease extent.
TACP Treatment (Transarterial Chemoperfusion) for stage 4 lung cancer in Germany
TACP Treatment (Transarterial Chemoperfusion) for stage 4 lung cancer in Germany is a regional infusion technique that delivers chemotherapy directly into tumor-supplying arteries without embolization. This approach focuses on increasing intratumoral drug concentration while maintaining vascular perfusion.
Under image guidance, a catheter is navigated into bronchial or pulmonary arterial branches feeding the tumor. Chemotherapy is infused at controlled rates to enhance tissue penetration. Because embolic agents are not used, arterial blood flow is preserved while achieving localized intensification of therapy.
Following infusion, vascular access is secured using standardized interventional methods. Patients are monitored for hemodynamic and respiratory stability. TACP may be considered in selected patients when regional tumor control is required in addition to systemic therapy. The cost of TACP Treatment in Germany typically ranges between €8,000 and €9,000 per session, depending on individual planning and disease extent.
Immunotherapy Dendritic cell therapy for stage 4 lung cancer in Germany
Immunotherapy Dendritic cell therapy for stage 4 lung cancer in Germany, is a personalized immune-based treatment strategy designed to activate tumor-specific T-cell responses. The process begins with comprehensive clinical evaluation within structured institutional programs.
A controlled blood collection isolates immune cells (Monocytes). These cells are transferred to an EU GMP certified laboratory, where they are cultured under regulated sterile conditions and differentiated into mature dendritic cells. Strict quality control procedures include sterility testing, viability assessment, and immune activation verification prior to release.
The final preparation is administered as a personalized dendritic cell vaccine, typically via intradermal injection at scheduled intervals. Clinical monitoring and imaging follow-up are performed to assess disease stability. This approach represents one of the innovative treatment options for NSCLC available within regulated oncology programs. The cost of Immunotherapy Dendritic Cell Therapy in Germany is €26,000 which includes laboratory processing and vaccine preparation.
Combining New Treatments for Non-Small Cell Lung Cancer Stage 4 in Germany
German oncology teams may evaluate combination strategies that include interventional treatments such as TPCE or TACP alongside immunotherapy. The goal is to address localized tumor activity while supporting systemic immune response. Patients are monitored closely through imaging and clinical assessment, and treatment plans are adapted based on response and tolerance.
New clinical trials and research for NSCLC in Germany
Germany actively supports oncology research and offers access to new clinical trials for NSCLC stage 4 exploring advanced treatments for non-small cell lung cancer stage 4. These trials investigate innovative drug combinations, immune-based therapies, and advanced interventional approaches under strict ethical oversight. Eligibility depends on tumor characteristics, prior treatments, and overall health.
Why Choose Treatment in Germany for Stage 4 Non–Small Cell Lung Cancer?
Choosing treatment within Germany’s healthcare system provides access to highly structured, multidisciplinary oncology programs designed specifically for complex cases such as stage 4 metastatic non-small cell lung cancer. Treatment is delivered through coordinated tumor boards that integrate medical oncology, interventional radiology, pathology, thoracic specialists, and immunotherapy teams, ensuring that all treatments options for stage 4 non–small cell lung cancer are evaluated comprehensively rather than in isolation.
Patients searching for the best oncologist in Germany benefit not only from individual physician expertise but from institutional treatment frameworks supported by latest medical technology in Germany, advanced interventional suites, and regulated cellular manufacturing facilities including an EU GMP certified laboratory. This infrastructure allows safe delivery of regional therapies such as TPCE and TACP, as well as immune-based approaches like a personalized dendritic cell vaccine.
Emphasis within German oncology centers is placed on regulatory compliance, treatment transparency, individualized planning, and long-term monitoring. Rather than fragmented care pathways, patients receive coordinated programs that integrate systemic therapy, regional interventional procedures, and innovative treatment options for NSCLC within a controlled and quality-assured medical environment
International Patient Support through Treatment in Germany
Access to advanced immunotherapy requires coordinated logistical planning in addition to clinical evaluation. Through Treatment in Germany (TIG), international patients can receive assistance with hospital coordination, appointment scheduling, travel arrangements, and medical visa support when required. Structured communication ensures that medical documentation is reviewed before travel and that treatment planning is individualized.
Treatment in Germany at www.treatmentingermany.de provides complete logistical support throughout the treatment pathway. Patients considering advanced immunotherapy option can get a free consultation with TIG (Treatment in Germany) to receive structured medical evaluation and coordinated guidance.
References
1-Howlader N et al. SEER Survival Statistics Review, 1975–2018. National Cancer Institute. SEER Program. Public Access
2-Khozin S et al. Real-World Outcomes of Patients With Advanced Non–Small Cell Lung Cancer. The Oncologist. PMC Free Article
3-Aguiar PN Jr et al. PD-L1 Expression in Non–Small Cell Lung Cancer: A Systematic Review and Meta-Analysis. Lung Cancer. PMC Free Article
4-SEER Cancer Stat Facts: Lung and Bronchus Cancer. National Cancer Institute. Public Access
5-Thai AA et al. Lung Cancer: Epidemiology, Biology, and Emerging Therapies. Journal of Clinical Medicine. Open Access
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:








.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)