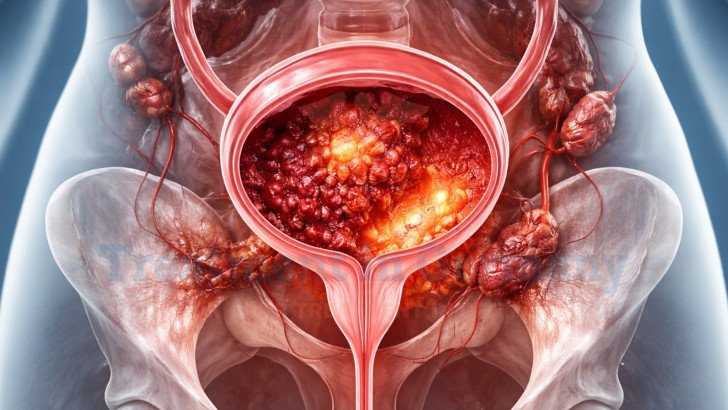
Stage 4 bladder cancer treatment in Germany including targeted embolization, dendritic cell vaccine therapy, innovative options, and coordinated support through TIG.
Stage 4 bladder cancer represents metastatic urothelial carcinoma that has extended beyond the bladder to regional lymph nodes or distant organs such as bladder cancer with liver metastases, lungs, or bones. According to Global Cancer Statistics 2020, bladder cancer accounted for 573,278 new cases and 212,536 deaths worldwide [1]. When diagnosed at a distant metastatic stage, outcomes are significantly reduced. Based on the most recent SEER Cancer Stat Facts data (2013–2019), the 5-year relative survival rate for distant-stage bladder cancer is 9% [2].
These figures highlight the aggressive biological behavior of advanced urothelial carcinoma and emphasize the importance of multidisciplinary management. In Germany, complex cases are typically evaluated within coordinated tumor boards that integrate systemic oncology, interventional radiology, and advanced cellular immunotherapy. Interventional oncology procedures, including Targeted embolization Treatment, are commonly associated with specialists such as Prof. Vogl, who has extensive experience in image-guided tumor embolization techniques.
In the field of immune-based therapies, Prof. Gansauge is recognized for his long-standing work in cellular immunotherapy development, including regulated laboratory preparation of personalized Immunotherapy Dendritic cell therapy strategies within an EU GMP certified laboratory environment. This combination of interventional and immune expertise supports access to innovative and individualized treatment approaches within structured oncology programs in Germany.
Muscle-invasive and advanced bladder cancers demonstrate frequent alterations in TP53 and RB1 tumor suppressor pathways, as well as dysregulation of PI3K/AKT and FGFR3 signaling, as reported by TCGA molecular profiling studies. [3].These genomic changes promote uncontrolled proliferation, invasion, and metastatic dissemination. Once cancer cells gain access to lymphatic channels or the bloodstream, they can colonize distant organs, leading to systemic disease.
The relatively high mutational burden characteristic of urothelial carcinoma contributes to immunogenic potential, which partly explains responsiveness to immune checkpoint inhibition. However, advanced disease also develops immune evasion strategies. Research has demonstrated that bladder cancer can impair dendritic cell maturation and reduce effective immune activation capacity [4]. The tumor microenvironment often contains suppressive immune cell populations that inhibit sustained anti-tumor responses. These mechanisms justify the integration of innovative immune-based approaches within selected comprehensive treatment strategies.
Platinum-based chemotherapy remains the standard first-line treatment for eligible patients with metastatic urothelial carcinoma. Combination regimens such as gemcitabine and cisplatin have demonstrated meaningful clinical benefit in advanced disease [5]. However, renal impairment and comorbidities limit eligibility in a substantial proportion of patients.
Immune checkpoint inhibitors have become established options following platinum failure. The phase III KEYNOTE-045 study confirmed improved overall survival with pembrolizumab compared with chemotherapy in previously treated advanced bladder cancer [6]. While immune checkpoint inhibition has improved outcomes for selected patients, progression remains common in widespread metastatic disease. Consequently, additional locoregional and immune-modulating strategies are explored within multidisciplinary oncology programs.
Targeted embolization Treatment for stage 4 bladder cancer, is a minimally invasive interventional oncology technique designed to selectively block the blood vessels supplying tumor tissue. In Stage 4 bladder cancer, this approach is most frequently considered for patients experiencing persistent tumor-related bleeding or for those with dominant metastatic lesions contributing significantly to symptom burden. Advanced urothelial carcinoma is often highly vascular, and tumor growth depends on continuous blood supply to maintain oxygenation and nutrient delivery. By selectively reducing this vascular support, embolization can induce controlled tumor ischemia and subsequent necrosis.
In cases of severe hematuria that are refractory to conservative measures, superselective vesical artery embolization has been shown to achieve effective hemorrhage control with acceptable safety outcomes [7]. This intervention can stabilize patients and significantly improve quality of life. The procedure is performed under image guidance, where a microcatheter is advanced into specific arterial branches supplying the tumor, allowing precise delivery of embolic material while preserving surrounding tissue.
Careful patient selection remains essential. In Germany, when clinically indicated, the cost of Targeted embolization Treatment ranges between €8,000–€9,000 per session. Treatment planning is individualized and conducted within multidisciplinary oncology programs.
Following confirmation of metastatic urothelial carcinoma, patients undergo comprehensive evaluation to determine suitability for Immunotherapy Dendritic cell therapy. Clinical history, prior systemic treatments, immune competence, organ function, and metastatic distribution are reviewed to ensure safe integration into the broader treatment strategy.
Once eligibility is confirmed, a controlled blood sample is collected to obtain specialized immune cells known as monocytes. These cells are isolated under sterile conditions and transferred to an EU GMP certified laboratory, where strict regulatory standards govern processing, sterility testing, and quality assurance. All laboratory procedures follow validated protocols designed to ensure safety, purity, and traceability.
Within controlled laboratory conditions, monocytes are cultured and differentiated into mature dendritic cells. During maturation, the cells are exposed to molecular signals derived from the patient’s tumor profile. This process results in a personalized dendritic cell vaccine designed to stimulate cytotoxic T lymphocytes capable of recognizing cancer cells.
Before clinical administration, the cellular product undergoes comprehensive quality-control testing, including sterility and viability assessment. Administration is typically performed intradermally. Early-phase clinical research has demonstrated feasibility and immune activation in patients receiving dendritic cell vaccination [8]. When clinically indicated, the cost of Immunotherapy Dendritic cell therapy in Germany is €26,000, reflecting immune cell collection, regulated laboratory processing, and individualized administration.
Ongoing Clinical Trials and New clinical trials for bladder cancer in Germany
International oncology guidelines recommend clinical trial participation for eligible patients with metastatic bladder cancer [5]. New clinical trials for bladder cancer in Germany investigate combination immunotherapy, antibody-drug conjugates, targeted molecular inhibitors, and cellular immunotherapy strategies.
Enrollment requires multidisciplinary evaluation and strict eligibility criteria. Participation may provide access to innovative therapeutic approaches under regulated supervision while contributing to advances in oncologic care.
Benefits and Clinical Considerations
Advanced interventions such as Targeted embolization Treatment offer localized symptom control and potential reduction in tumor burden. Immunotherapy Dendritic cell therapy provides a personalized immune activation strategy designed to complement systemic regimens. These approaches represent innovative additions to comprehensive stage 4 bladder cancer management in Germany.
However, therapeutic response varies depending on metastatic burden, molecular subtype, immune status, and prior therapy exposure. Careful multidisciplinary evaluation remains essential to balance potential benefits with realistic clinical expectations.
Why Choose Germany for Advanced Stage 4 Bladder Cancer Treatment?
Germany provides a highly regulated clinical and laboratory framework for managing Stage 4 bladder cancer, integrating systemic oncology, interventional radiology, and cellular immunotherapy within structured multidisciplinary programs. Treatment planning in specialized German oncology centers involves coordinated evaluation by medical oncologists, interventional radiologists, urologic surgeons, and immunotherapy specialists. This collaborative model ensures that complex metastatic cases are assessed comprehensively before individualized therapy decisions are made.
Advanced interventional procedures such as Targeted embolization Treatment are performed using precision-guided imaging systems that support safe vascular access and selective tumor targeting. In parallel, immune-based approaches such as Immunotherapy Dendritic cell therapy are developed within an EU GMP certified laboratory, where validated manufacturing protocols ensure strict compliance with sterility, quality assurance, and traceability standards. This regulated infrastructure is essential for advanced cellular therapy preparation.
Access to latest medical technology in Germany supports accurate staging, treatment monitoring, and procedural precision. Structured tumor board discussions allow individualized evaluation of metastatic distribution, prior therapy response, and overall performance status. These elements collectively contribute to expanding new and innovative treatment options in Germany, offering carefully implemented, evidence-based strategies for advanced bladder cancer management.
International Patient Support through Treatment in Germany
Access to advanced management for stage 4 bladder cancer involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as Targeted embolization Treatment or Immunotherapy Dendritic cell therapy. Through Treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This preliminary evaluation helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and Innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
1- Sung H et al. Global Cancer Statistics 2020. CA Cancer J Clin. Open Access.
2- SEER Cancer Stat Facts: Bladder Cancer. National Cancer Institute. Public Registry (2013–2019).
3- Robertson AG et al. Comprehensive Molecular Characterization of Muscle-Invasive Bladder Cancer. Cell. Open Access.
4- Xiu W et al. Bladder Cancer Suppresses Dendritic Cell Function. Oncology Letters. PMC Free Article.
5- von der Maase H et al. Gemcitabine and Cisplatin vs MVAC in Advanced Bladder Cancer. J Clin Oncol. Open Access.
6- Bellmunt J et al. Pembrolizumab vs Chemotherapy (KEYNOTE-045). J Clin Oncol. PMC Free Article.
7- Saadi A et al. Superselective Vesical Artery Embolization. Case Reports in Urology. PMC Free Article.
8- Maeng HM et al. Phase I Study of Dendritic Cell Vaccine. J Immunol Res. PMC Free Article.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:
Stage 4 bladder cancer is advanced disease that has spread beyond the bladder to distant organs such as lymph nodes, liver, lungs, or bones.
Common causes include smoking, chemical exposure, chronic bladder irritation, age, and genetic factors.
Blood in urine, frequent urination, urgency, and burning during urination are common early signs.
One innovative approach includes combining systemic therapy with Immunotherapy Dendritic cell therapy and Targeted embolization Treatment in selected cases.
It is a minimally invasive procedure that blocks blood flow to tumor tissue to control bleeding or reduce tumor burden.
A Dendritic cell vaccine activates the patient’s immune system by stimulating T cells to recognize and attack bladder cancer cells.
The Side effects of dendritic cell therapy are generally mild and may include temporary flu-like symptoms or localized injection-site reactions.
Yes. It may be integrated with systemic chemotherapy depending on clinical evaluation and overall treatment strategy.
Side effects vary but are monitored closely in cancer treatment in Germany to reduce discomfort.
Yes, some patients may be evaluated for new clinical trials, depending on eligibility criteria.
International patients can contact TIG, (Treatment in Germany) at www.treatmentingermany.de which manages complete logistical arrangements including a medical visa support if required.
Kindly complete the form below, and our dedicated team will reach out to you promptly. We look forward to connecting with you soon!
Trierer Straße, 56072 Koblenz, Germany

.webp)
 (1).webp)

.webp)
 (1).webp)


.webp)
 (1).webp)

.webp)
 (1).webp)
