Learn about glioblastoma treatment in Germany, including dendritic cell immunotherapy, survival data, symptoms, and new clinical trials for WHO grade 4 brain tumors.
Glioblastoma Treatment in Germany – Dendritic Cell Therapy for WHO Grade 4 Brain Tumor
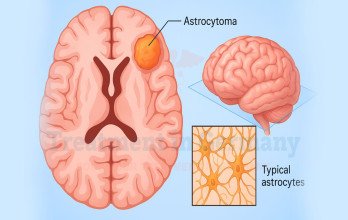
Glioblastoma is the most aggressive primary malignant brain tumor in adults and represents the highest grade within diffuse astrocytic tumors. It originates from astrocytes and demonstrates rapid cellular proliferation, extensive infiltration into surrounding brain tissue, and resistance to standard therapies. Clinically, it is classified as a WHO grade 4 brain tumor, a designation that reflects its highly malignant biological behavior. Many patients refer to it as level 4 glioblastoma brain cancer, which corresponds to the same diagnostic classification.
Modern neuropathology distinguishes most adult glioblastomas as IDH-wildtype glioblastoma, a molecular subtype defined by the absence of IDH mutations and associated with more aggressive progression. The 2021 WHO Classification of Tumors of the Central Nervous System formally incorporates molecular features into diagnostic criteria, emphasizing genomic profiling in treatment planning [1]. Because of the tumor’s severity and high recurrence risk, patients frequently search for the best glioblastoma treatment in the world soon after diagnosis.
Within structured neuro-oncology programs in Germany, treatment decisions are made by multidisciplinary tumor boards integrating neurosurgery, radiation oncology, neuropathology, and immune-based therapy expertise. Surgical intervention is performed by experienced neuro surgeon teams, while advanced immune strategies such as dendritic cell therapy are implemented within regulated institutional programs. This approach is supported by specialist-led immunotherapy programs in Germany, including those guided by highly experienced experts such as Prof. Gansauge, known for his focused work in advanced cell-based cancer immunotherapy.
Symptoms of Glioblastoma and Clinical Presentation
The symptoms of glioblastoma depend on tumor size and location within the brain. Because the tumor grows rapidly and infiltrates adjacent tissue, symptoms may develop over weeks to months. Common manifestations include persistent headaches, seizures, progressive neurological deficits, cognitive decline, personality changes, and weakness affecting one side of the body. Increased intracranial pressure may also cause nausea, vomiting, or visual disturbances.
Neurological examination findings often guide urgent imaging evaluation. Magnetic resonance imaging typically demonstrates a contrast-enhancing mass with surrounding edema and central necrosis. Due to the infiltrative nature of glioblastoma, tumor margins extend microscopically beyond visible imaging boundaries. These characteristics contribute to recurrence despite maximal surgical resection.
Survival Outcomes in IDH-Wildtype Glioblastoma
Despite multimodal therapy, survival outcomes remain limited. A randomized clinical trial comparing radiotherapy alone with radiotherapy plus temozolomide demonstrated that median overall survival improved from 12.1 months to 14.6 months when chemotherapy was added [2]. This combined chemoradiotherapy protocol remains the foundation of modern standard treatment.
Long-term follow-up of the same study confirmed sustained survival benefit with the combined approach [3]. Population-based analyses further indicate that overall survival remains limited in routine clinical practice [4]. These findings explain why recurrence is common and why many patients explore glioblastoma recurrence treatment options and glioblastoma experimental treatment strategies after standard therapy.
Why Glioblastoma Is Biologically Aggressive
The aggressive behavior of IDH-wildtype glioblastoma is closely linked to its molecular landscape. Comprehensive genomic analyses have identified frequent alterations such as EGFR amplification and TERT promoter mutations, which promote uncontrolled cellular growth and resistance to cytotoxic therapy [5]. These molecular features drive rapid tumor progression and limit responsiveness to conventional treatments.
Glioblastoma also establishes a profoundly immunosuppressive tumor microenvironment. Tumor-associated macrophages and regulatory immune cells inhibit effective cytotoxic T-cell responses, weakening natural immune surveillance [6]. In addition, the blood–brain barrier restricts penetration of systemic therapies into infiltrative tumor regions. These biological barriers justify investigation into new and innovative treatments for glioblastoma in Germany, particularly immune-based approaches.
Standard Treatment for Glioblastoma in Germany
Initial treatment for newly diagnosed glioblastoma includes maximal safe surgical resection performed by a specialized neuro surgeon, followed by radiotherapy combined with temozolomide chemotherapy. Surgical resection aims to reduce tumor mass while preserving neurological function. However, due to microscopic infiltration into surrounding brain tissue, complete removal is not possible.
Radiotherapy targets residual tumor cells, while temozolomide enhances cytotoxic effects during and after radiation. Although this multimodal approach improves survival compared to radiotherapy alone, recurrence remains common.
Within Germany’s healthcare system, structured tumor boards integrate surgical, medical, and immune-based strategies. Treatment planning emphasizes safety, regulatory compliance, and individualized assessment rather than isolated consultation.
Dendritic Cell Therapy in Germany: Personalized Immunotherapy for Glioblastoma
Immunotherapy for glioblastoma aims to activate the patient’s immune system to recognize and attack tumor cells. Among immune-based strategies, dendritic cell therapy represents a personalized approach designed to stimulate tumor-specific T lymphocyte responses.
Dendritic cells are professional antigen-presenting cells responsible for initiating adaptive immune responses. In glioblastoma, tumor-induced immune suppression interferes with effective antigen presentation, reducing natural anti-tumor activity. Dendritic cell therapy attempts to overcome this suppression by externally activating dendritic cells and reintroducing them in a primed state capable of stimulating cytotoxic T cells. This strategy is considered part of new and innovative treatments for glioblastoma in Germany, delivered within structured institutional programs at recognized centers.
How Dendritic Cell Therapy Is Performed in Germany
The treatment process begins with comprehensive evaluation in a multidisciplinary tumor board. Patients with level 4 glioblastoma brain cancer, particularly those diagnosed with IDH-wildtype glioblastoma, undergo assessment of imaging findings, prior therapy exposure, and neurological function to determine eligibility.
A controlled blood collection is performed to isolate monocytes, which serve as immune cells (monocytes). These cells are transported to an EU GMP certified laboratory, where cellular manufacturing occurs under validated sterile conditions. Monocytes are cultured with specific cytokines that induce differentiation into mature dendritic cells capable of activating tumor-specific T lymphocytes.
Following maturation, dendritic cells are exposed to tumor-derived lysate material prepared from surgical specimens. This exposure allows dendritic cells to process tumor-associated molecular signals and prepare them for immune presentation. Rigorous quality control procedures are performed before release, including sterility testing, endotoxin screening, viability assessment, and phenotypic confirmation of immune activation markers.
The prepared dendritic cells vaccine is administered via intradermal injections according to a structured schedule. Patients are monitored for immune responses and clinical stability. Imaging follow-up assesses disease status over time. Programs operate using latest medical technology in Germany, ensuring documentation, traceability, and regulatory compliance. The cost of Immunotherapy Dendritic Cell Therapy in Germany is €26,000 which includes laboratory processing and vaccine preparation.
Clinical Research and Ongoing Trials
A Phase III externally controlled cohort study evaluating autologous tumor lysate–loaded dendritic cell vaccination demonstrated prolonged overall survival compared with matched control populations [7]. Additional studies have documented measurable immune activation following vaccination, including increased tumor-specific T-cell responses [8]. These findings support continued investigation within structured programs and new clinical trials for glioblastoma in Germany designed to evaluate advanced immune strategies.
Limitations and Considerations of Treatment in Germany
Although dendritic cell therapy represents an advanced immune-based strategy, careful patient selection remains essential. Not all individuals with WHO grade 4 brain tumor are appropriate candidates, and therapeutic response may vary depending on tumor burden, molecular profile, and immune status. Cases of IDH-wildtype glioblastoma often demonstrate aggressive progression despite intensive therapy.
Cellular manufacturing requires specialized infrastructure and strict regulatory oversight. The necessity of an EU GMP certified laboratory ensures safety and quality but limits availability to specialized centers. Immune-based therapy is typically integrated with surgery, radiation, or chemotherapy rather than replacing standard modalities. Patients seeking the best glioblastoma treatment in the world should understand that innovative therapies complement comprehensive neuro-oncology programs and require multidisciplinary coordination.
Why Choose Treatment in Germany for Glioblastoma
Patients searching for the best oncologist in Germany benefit from structured institutional programs within Germany’s healthcare system, where neurosurgery, molecular diagnostics, radiation oncology, and immune-based therapies are integrated through coordinated tumor boards.
Access to new clinical trials for glioblastoma in Germany expands treatment possibilities for eligible patients. Recognized centers maintain rigorous laboratory standards, quality control measures, and regulatory compliance. International patients can receive coordinated medical planning through TIG (Treatment in Germany), ensuring safe access to advanced care.
International Patient Support through Treatment in Germany
Access to advanced management for glioblastoma involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as Immunotherapy Dendritic cell therapy. Through Treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This pre-evaluation by the treating physician helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and Innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
References
1- Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, et al. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathologica. 2021;142(3):481–498. Open Access.
2- Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJB, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. New England Journal of Medicine. 2005;352(10):987–996. PMC Free Article.
3- Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJB, Janzer RC, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma: long-term analysis of the EORTC-NCIC trial. Lancet Oncology. 2009;10(5):459–466. PMC Free Article.
4- Ostrom QT, Gittleman H, Xu J, Kromer C, Wolinsky Y, Kruchko C, et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States. Neuro-Oncology. 2019;21(Suppl 5):v1–v100. PMC Free Article.
5- Brennan CW, Verhaak RGW, McKenna A, Campos B, Noushmehr H, Salama SR, et al. The somatic genomic landscape of glioblastoma. Cell. 2013;155(2):462–477. Open Access.
6- Lim M, Xia Y, Bettegowda C, Weller M. Current state of immunotherapy for glioblastoma. Nature Reviews Clinical Oncology. 2018;15(7):422–442. PMC Free Article.
7- Liau LM, Ashkan K, Tran DD, Campian JL, Trusheim JE, Cobbs CS, et al. First results on survival from a large Phase III trial of an autologous dendritic cell vaccine in newly diagnosed glioblastoma. JAMA Oncology. 2023;9(1):112–121. PMC Free Article.
8- Batich KA, Reap EA, Archer GE, Sanchez-Perez L, Nair SK, Schmittling RJ, et al. Long-term survival in glioblastoma with cytomegalovirus pp65-targeted vaccination. Clinical Cancer Research. 2017;23(8):1898–1909. PMC Free Article.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:






.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)