Comprehensive guide to Stage 4 liver cancer treatment in Germany, including TACE Treatment, Immunotherapy Dendritic Cell Therapy, symptoms, and complete logistical arrangements through TIG (Treatment in Germany).
Stage 4 Liver Cancer: Full Guide and Treatment Options in Germany
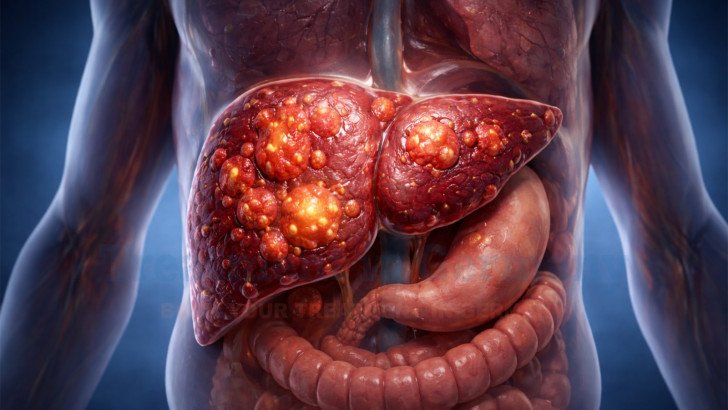
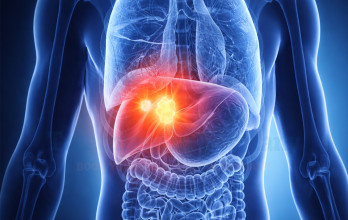
Stage 4 liver cancer represents the most advanced stage of hepatocellular carcinoma, the most common primary malignancy of the liver. At this stage, the tumor has spread beyond the liver or involves distant organs, a condition often described as metastasized liver cancer. Advanced disease significantly limits surgical options and requires comprehensive treatment planning. Management must balance tumor control with preservation of hepatic function, particularly because many patients have underlying cirrhosis or chronic viral hepatitis.
Liver cancer continues to be a major global health burden. Global Cancer Statistics 2020 reported 905,677 new cases of liver cancer and 830,180 deaths worldwide in 2020 [1]. These figures reflect the aggressive biological behavior of hepatocellular carcinoma and the high mortality associated with late-stage presentation. Because stage 4 disease frequently presents with extrahepatic spread, patients often search for new and innovative treatment options for liver cancer that extend beyond conventional systemic therapy.
Within Germany’s healthcare system, advanced liver cancer cases are evaluated in multidisciplinary tumor boards integrating oncology, hepatology, and interventional radiology. Interventional procedures such as TACE Treatment are performed by experienced specialists including Prof. Vogl, while immune-based strategies such as Immunotherapy Dendritic Cell Therapy are implemented within regulated institutional programs guided by Prof. Gansauge. This structured environment ensures individualized planning rather than isolated decision-making.
Understanding Stage 4 and Metalised Liver Cancer
Stage 4 liver cancer may involve multiple tumors within the liver or metastases to distant organs. The term metastasized liver cancer reflects systemic spread beyond hepatic tissue. Advanced liver cancer often develops in the setting of cirrhosis, meaning patients face both malignant progression and compromised liver function.
Tumor invasion disrupts bile flow, metabolic pathways, and coagulation mechanisms. When metastases occur, additional organ systems may be affected. Careful staging using contrast-enhanced imaging allows physicians to assess tumor distribution and determine eligibility for metastatic liver cancer treatment strategies such as embolization-based therapy.
Laboratory evaluation includes bilirubin levels, albumin concentration, and coagulation parameters. These measurements help determine hepatic reserve and guide therapeutic decision-making. Because advanced liver cancer affects systemic physiology, treatment must be carefully tailored.
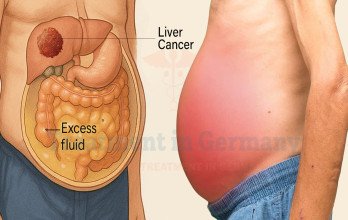
Symptoms of Stage 4 Liver Cancer
The latest symptoms of liver cancer in advanced stages include fatigue, weight loss, right upper abdominal pain, and jaundice. Fluid accumulation in the abdomen may cause swelling and discomfort. Reduced appetite and early satiety are also common as tumor burden increases.
When disease progresses to metastasized liver cancer, symptoms may reflect metastatic involvement. Pulmonary metastases can cause shortness of breath, while bone metastases may produce localized pain. Because hepatic function is essential for metabolism and detoxification, tumor progression often leads to systemic decline.
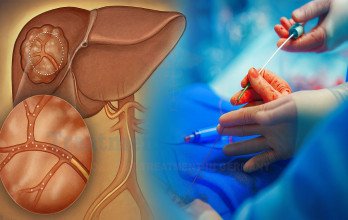
TACE Treatment for Advanced Liver Cancer in Germany
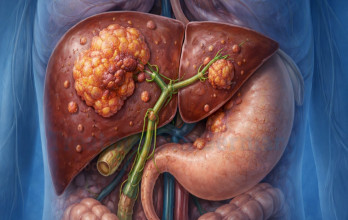
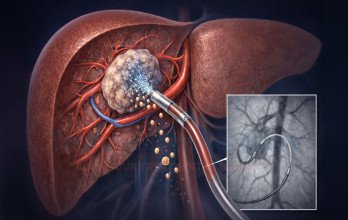
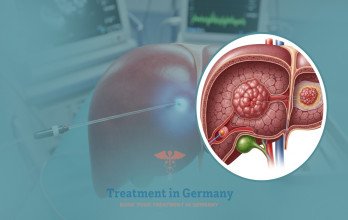
TACE Treatment for advanced liver cancer is a minimally invasive interventional oncology procedure commonly used in patients whose tumor burden remains predominantly confined to the liver. The therapy delivers chemotherapy directly into tumor-feeding vessels, followed by embolic material that blocks the blood flow to malignant tissue. This dual mechanism enhances local cytotoxic effect while limiting systemic exposure.
The vascular supply of hepatocellular carcinoma enables selective targeting through catheter-based techniques. Under imaging guidance, a catheter is introduced through a peripheral artery and navigated to tumor-feeding vessels. Chemotherapy is infused, followed by embolization particles that blocks the blood flow, promoting tumor necrosis.
The procedure is performed in advanced angiography suites using precision imaging technology. Post-procedural monitoring ensures early management of temporary symptoms such as mild fever or abdominal discomfort. Multidisciplinary tumor boards determine whether TACE should be used as monotherapy or combined with systemic treatment. The cost of TACE therapy in Germany typically ranges between €8,000 and €9,000 per session.
Immunotherapy Dendritic Cell Therapy for advanced liver cancer in Germany
Immunotherapy Dendritic Cell Therapy is a personalized immune-based treatment approach designed to stimulate a targeted anti-tumor response in patients with advanced liver cancer. In liver cancer, the tumor microenvironment often suppresses immune activity, reducing the effectiveness of cytotoxic T lymphocytes that would otherwise recognize and destroy malignant cells. Dendritic cell therapy aims to reverse this immune suppression by activating the patient’s own immune system in a controlled manner. By enhancing immune recognition of cancer cells, this strategy seeks to support systemic anti-tumor activity as part of a comprehensive treatment plan.
The treatment process begins with a detailed clinical evaluation to determine eligibility based on liver function, tumor burden, and overall health status. Once approved, a controlled blood sample is collected to isolate immune cells (Monocytes). These cells are transported to an EU GMP certified laboratory, where they are cultured under sterile, validated conditions. During laboratory processing, monocytes are differentiated into mature dendritic cells and exposed to tumor-derived material obtained from the patient. This step enables the cells to acquire the functional ability to activate tumor-specific T lymphocytes after re-administration.
Before clinical use, the prepared cellular product undergoes strict quality control testing to ensure sterility, viability, and regulatory compliance. The matured dendritic cells are then administered through intradermal injections according to a structured schedule. Patients are monitored through follow-up imaging and laboratory assessments to evaluate tolerance and clinical response. Growing interest in the immunotherapy liver cancer success rate reflects continued investigation into immune-based strategies for advanced hepatic malignancies. The cost of Immunotherapy Dendritic Cell Therapy in Germany is €26,000 which includes laboratory processing and vaccine preparation.
New and ongoing clinical trials for advanced liver cancer in Germany
Ongoing clinical research continues to explore the integration of immunotherapy with locoregional and systemic approaches in advanced hepatocellular carcinoma. Contemporary scientific literature highlights the growing interest in immune-based strategies that aim to enhance anti-tumor responses while maintaining hepatic stability. Combination approaches that integrate interventional procedures with immune modulation are being investigated to improve disease control in patients with advanced and metastatic involvement. These developments contribute to the evolution of new and innovative treatment options for liver cancer, particularly in specialized oncology centers.
Patients with advanced disease may be evaluated for participation in new clinical trials for liver cancer in Germany, depending on individual eligibility criteria and institutional availability. Clinical research programs are conducted within regulated frameworks to ensure patient safety, ethical oversight, and scientific rigor. Participation in structured research initiatives may provide access to emerging therapeutic strategies while contributing to broader advancements in liver cancer management.
Limitations and Considerations of Treatment in Germany
Stage 4 liver cancer management requires thorough evaluation of both tumor extent and underlying liver function before any therapeutic decision is made. Because many patients with advanced hepatocellular carcinoma have cirrhosis or chronic liver disease, hepatic reserve must be carefully assessed to avoid treatment-related decompensation. Locoregional approaches such as TACE Treatment may not be suitable for individuals with severely impaired liver function or widespread extrahepatic metastases. In cases of metalised liver cancer, therapy must address both intrahepatic tumor burden and distant spread, which often requires integration of systemic strategies.
Multidisciplinary tumor boards within Germany’s healthcare system evaluate imaging findings, laboratory results, and overall performance status to ensure that selected metastatic liver cancer treatment options are clinically appropriate and safe. Immune-based approaches such as Immunotherapy Dendritic Cell Therapy also require individualized assessment. The effectiveness of immune activation depends on tumor biology, immune competence, and prior treatment exposure.
Not all patients respond equally, and outcomes may vary depending on disease distribution and systemic condition. Cellular therapy must be manufactured within an EU GMP certified laboratory, where strict regulatory standards ensure sterility, quality control, and traceability. While these safeguards enhance patient safety, they limit availability to specialized centers with validated infrastructure. For this reason, innovative therapies are integrated into comprehensive oncologic planning rather than used in isolation, ensuring coordinated care within structured treatment programs in Germany.
Why Choose Treatment in Germany
Germany offers structured multidisciplinary oncology programs integrating hepatology, interventional radiology, and immunotherapy expertise. Access to latest medical technology in Germany enhances procedural precision and patient monitoring. Regulated laboratory environments ensure standardized manufacturing processes for cellular therapies.
International patients receive coordinated support through TIG (Treatment in Germany), which facilitates documentation review, hospital coordination, appointment scheduling, and travel planning. This organized framework enables safe access to advanced liver cancer treatment programs.
International Patient Support through Treatment in Germany
Access to advanced management for liver cancer involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as TACE Treatment or Immunotherapy Dendritic cell therapy through Treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This pre-evaluation by the treating physician helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and Innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
References
1- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020. CA Cancer J Clin. 2021;71(3):209–249. doi:10.3322/caac.21660. Open Access.
2- Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2019;16:589–604. doi:10.1038/s41575-019-0186-y. Open Access.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:
















.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)