Bile Duct Cancer (Cholangiocarcinoma) treatment in Germany including dendritic cell therapy, TACE therapy, and complete logistical arrangements through TIG.
Bile Duct Cancer (Cholangiocarcinoma): Modern Treatment Options in Germany
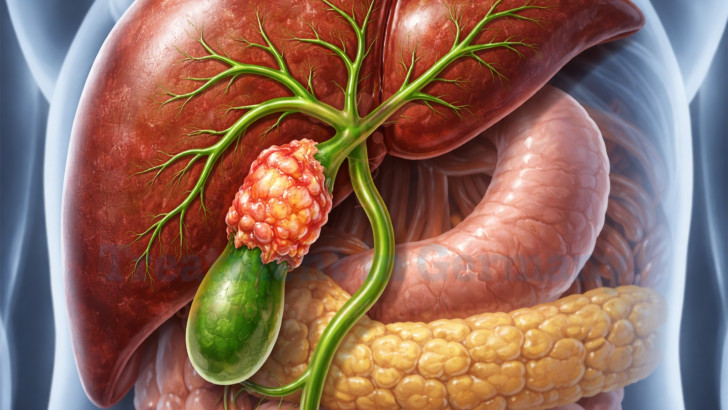
Bile duct cancer, medically known as cholangiocarcinoma, is a rare but aggressive malignancy arising from the epithelial lining of the intrahepatic or extrahepatic bile ducts. According to Global Cancer Statistics 2022, liver and intrahepatic bile duct cancers accounted for 905,677 new cases and 830,180 deaths worldwide in 2022 [1]. Cholangiocarcinoma represents a major proportion of biliary tract cancers and contributes significantly to the global burden of hepatobiliary malignancies. The incidence of cholangiocarcinoma has shown geographic variation, with higher rates reported in certain regions of Asia and increasing trends observed in Western countries [2].
Data from the SEER Cancer Stat Facts indicate that the 5-year relative survival rate for bile duct cancer diagnosed between 2013 and 2019 was 10.5 percent overall, with survival strongly dependent on stage at diagnosis [3]. Many patients present with advanced disease due to nonspecific early symptoms such as jaundice, abdominal discomfort, or weight loss. As a result, comprehensive staging and individualized treatment planning are essential.
Within Germany’s healthcare system, patients with cholangiocarcinoma are treated in certified German healthcare centers operating under structured oncology programs in Germany. Cases are reviewed in multidisciplinary tumor board meetings involving hepatobiliary surgeons, leading medical oncologists, interventional radiologists, and oncology specialists. Prof. Vogl is recognized as an expert in image-guided interventional oncology and embolization therapies, while Prof. Gansauge specializes in regulated cellular immunotherapy programs, including Immunotherapy Dendritic Cell Therapy. This coordinated approach enables access to comprehensive and innovative bile duct cancer treatment in Germany.
Understanding How Bile Duct Cancer (Cholangiocarcinoma) Develops and Progresses
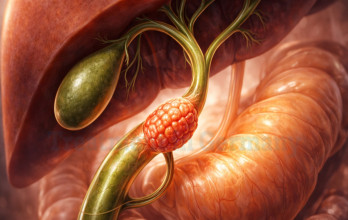
Cholangiocarcinoma develops from the epithelial cells lining the bile ducts, which transport bile from the liver to the small intestine. Chronic inflammation of the bile ducts, primary sclerosing cholangitis, bile duct cysts, and certain parasitic infections are recognized risk factors. Genetic alterations affecting cell proliferation and DNA repair pathways contribute to malignant transformation.
The disease is categorized anatomically into intrahepatic, perihilar, and distal bile duct cancers, each with distinct clinical and surgical implications. Tumor growth may obstruct bile flow, leading to jaundice and liver dysfunction. In advanced stages, cancer can invade surrounding vascular structures or metastasize to lymph nodes and distant organs.
Accurate staging requires advanced diagnostics, including cross-sectional imaging and laboratory evaluation to assess liver function and tumor markers. Imaging findings guide surgical planning, systemic therapy decisions, and eligibility for regional interventional procedures such as TACE therapy.
Standard Systemic Therapy and Clinical Management
Surgical resection offers the only potentially curative option for localized cholangiocarcinoma. However, many patients are diagnosed at advanced stages when complete resection is not feasible. In such cases, systemic chemotherapy becomes the cornerstone of treatment, commonly using gemcitabine-based combination regimens.
For unresectable or metastatic disease, systemic therapy aims to control tumor growth and preserve quality of life. Targeted therapies may be considered in patients with specific molecular alterations identified through genomic profiling. Radiation therapy can be used selectively for local control or palliation. Management decisions are made within multidisciplinary tumor board discussions in structured oncology programs in Germany. Continuous monitoring through imaging and laboratory assessment allows timely adaptation of treatment strategy. Personalized treatment planning is central to comprehensive bile duct cancer treatment in Germany.
Dendritic Cell Therapy for Bile Duct Cancer in Germany
Dendritic cell therapy for bile duct cancer represents a personalized immunotherapeutic strategy integrated into selected cases within regulated programs. The process begins with comprehensive medical evaluation, including tumor characterization, immune assessment, and multidisciplinary tumor board review to determine eligibility within structured oncology programs in Germany.
Once approved, the procedure starts with collection of immune cells (monocytes) from the patient’s blood under controlled clinical conditions. These cells are transferred to an EU GMP certified laboratory operating under strict regulatory standards. In the laboratory, monocytes are differentiated and matured into dendritic cells capable of presenting tumor-associated antigens.
The cells are then exposed to tumor-derived antigens specific to the patient’s cholangiocarcinoma profile, resulting in a personalized dendritic cell vaccine. Before administration, the product undergoes sterility testing, viability analysis, and quality control verification. The vaccine is administered intradermally according to a structured schedule, followed by immune and clinical monitoring. The cost of Dendritic Cell Therapy in Germany is €27,000. This therapy remains investigational and is considered in highly selected patients within regulated clinical programs.
TACE Therapy for Bile Duct Cancer in Germany
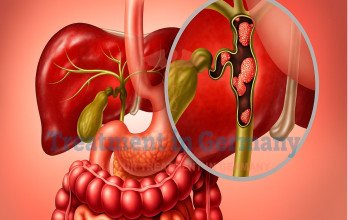
TACE therapy for bile duct cancer may be considered in selected patients with unresectable intrahepatic cholangiocarcinoma or dominant liver involvement. In advanced disease, tumor growth within the liver can significantly influence prognosis and symptom burden. TACE delivers concentrated chemotherapy directly into the blood vessels supplying the tumor while blocking the blood flow to enhance local therapeutic effect.
The procedure is performed under real-time imaging guidance in specialized interventional oncology units within certified German healthcare centers equipped with the latest medical technology in Germany. A catheter is inserted through a small arterial access point, typically in the groin, and guided toward the vessels feeding the tumor. Once selective positioning is confirmed, chemotherapy is infused directly into the targeted branches, followed by embolic material that blocks tumor blood supply and prolongs drug contact within cancer tissue.
After TACE therapy, structured follow-up includes liver function assessment and imaging reassessment to evaluate radiologic response. The procedure may be integrated with systemic chemotherapy depending on overall disease distribution and multidisciplinary tumor board recommendations. The cost of TACE therapy in Germany typically ranges between €8,000 and €9,000 per session, delivered within regulated cancer treatment programs.
Ongoing Clinical Research and New Clinical Trials in Germany
German oncology institutions actively participate in research exploring targeted therapies, immunotherapy combinations, and cellular-based treatments for cholangiocarcinoma. Eligible patients may access new clinical trials for bile duct cancer after molecular profiling and structured evaluation.
Research is conducted within regulated frameworks under Germany’s healthcare system, ensuring ethical oversight and patient safety. Collaboration with international organizations, including the Canadian Cancer Society, supports global knowledge exchange and scientific advancement.
Enrollment in clinical trials is reviewed by multidisciplinary tumor boards within recognized centers considered among the best bile duct cancer treatment centers. These programs aim to expand access to new and innovative treatment options for bile duct cancer while maintaining responsible clinical standards.
Limitations and Considerations for Bile Duct Cancer
Cholangiocarcinoma often presents at an advanced stage, and treatment outcomes depend on tumor location, extent of spread, liver function, and overall patient health. While surgery offers potential curative intent in localized cases, many patients require systemic or regional therapies.
Interventional procedures such as TACE therapy are appropriate for selected patients and are not substitutes for systemic treatment in widely metastatic disease. Dendritic cell therapy requires careful immune evaluation and structured follow-up within regulated programs.
Individualized planning through multidisciplinary tumor board discussion remains essential. Ongoing monitoring and adaptive treatment strategies support balanced integration of advanced therapies into comprehensive bile duct cancer management.
Why Choose Treatment for Bile Duct Cancer in Germany?
Cancer treatment in Germany is delivered through certified German healthcare centers operating under strict regulatory standards within Germany’s healthcare system. These institutions integrate hepatobiliary surgery, systemic oncology, interventional radiology, and cellular immunotherapy within structured oncology programs in Germany.
Access to the latest medical technology in Germany supports precise diagnostics and minimally invasive interventional procedures. Multidisciplinary tumor board evaluation ensures that each treatment plan aligns with clinical guidelines and individual patient factors. For international patients, bile duct cancer treatment in Germany offers coordinated treatment pathways, experienced oncology specialists, and access to regulated and innovative therapeutic options.
International Patient Support through Treatment in Germany (TIG)
Access to advanced management for bile duct cancer involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as Immunotherapy Dendritic cell and TACE therapy, through treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This preliminary evaluation by the treating physician helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
References
1- Bray F, Laversanne M, Sung H, et al. Global Cancer Statistics 2022. CA Cancer J Clin. 2024. Open Access.
2- Banales JM, Marin JJG, Lamarca A, et al. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol. 2020. PMC Free Article.
3-National Cancer Institute. SEER Cancer Stat Facts: Bile Duct Cancer. Public Registry.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:




.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)