Triple-Negative Breast Cancer treatment in Germany including dendritic cell therapy, TACE, TACP, and complete logistical support through TIG.
Triple-Negative Breast Cancer: Latest Treatment Options in Germany
Triple-negative breast cancer, commonly referred to as TNBC, is a biologically distinct subtype defined by the absence of estrogen receptors, progesterone receptors, and HER2 expression. According to Global Cancer Statistics 2020, female breast cancer accounted for 2,261,419 new cases and 684,996 deaths worldwide in 2020 [1]. TNBC represents approximately 10 to 15 percent of all breast cancers and is associated with specific molecular characteristics and a higher likelihood of early recurrence compared with hormone receptor positive disease [2].
The SEER Cancer Stat Facts report that the 5-year relative survival rate for female breast cancer diagnosed between 2013 and 2019 was 90.8 percent overall, with survival varying significantly according to stage at diagnosis [3]. TNBC is more frequently diagnosed in younger women and may be associated with inherited BRCA1 mutations [2]. Because endocrine and HER2-targeted therapies are not effective in this subtype, systemic chemotherapy and immunotherapy form the backbone of treatment.
Within Germany’s healthcare system, patients receive care in certified cancer centers operating under structured oncology programs. All complex cases are reviewed by a multidisciplinary tumor board to ensure individualized treatment planning. Prof. Vogl is recognized as an expert in image-guided interventional oncology and embolization therapies, while Prof. Gansauge specializes in regulated cellular immunotherapy programs, including Immunotherapy Dendritic Cell Therapy. This coordinated framework enables access to comprehensive Triple-Negative Breast Cancer treatment delivered by experienced oncology specialists.
Understanding How Triple-Negative Breast Cancer Develops and Progresses
TNBC develops when genetic alterations in breast epithelial cells result in uncontrolled growth and loss of receptor expression. These tumors often demonstrate high proliferative activity and genomic instability, contributing to faster progression compared with other breast cancer subtypes. The absence of hormone-driven pathways shifts therapeutic strategies toward cytotoxic and immune-based treatments.
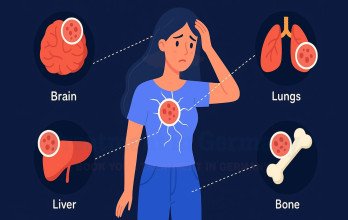
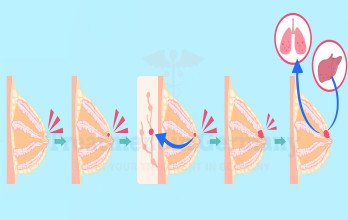
Disease progression may involve regional lymph nodes and distant organs such as TNBC with liver metastases, lungs, brain, or bones. Accurate staging relies on advanced diagnostics to determine tumor burden and metastatic spread. Imaging findings guide decisions regarding surgery, systemic therapy, and potential interventional procedures. Biological heterogeneity is a defining feature of TNBC. Some tumors display immune-rich microenvironments, which may influence response to immunotherapy, while others show DNA repair deficiencies relevant for targeted agents. Comprehensive evaluation ensures treatment aligns with tumor biology and patient-specific clinical factors.
Standard Systemic Therapy and Clinical Management
For early-stage TNBC, treatment typically includes surgery combined with chemotherapy. Many patients receive neoadjuvant chemotherapy to reduce tumor size before surgery and to evaluate pathological response. Regimens commonly include anthracyclines and taxanes, with platinum agents considered in selected cases based on molecular features.
In metastatic TNBC, systemic chemotherapy remains central to disease control. Immune checkpoint inhibitors may be combined with chemotherapy in appropriately selected patients [2]. PARP inhibitors can be considered in individuals with germline BRCA mutations. Radiation therapy may be recommended following breast-conserving surgery or for symptom control in advanced disease.
Treatment decisions are individualized and guided by multidisciplinary tumor board discussion. Continuous clinical assessment and imaging follow-up support timely adaptation of therapy. German oncology centers emphasize evidence-based care and access to innovative therapeutic strategies when clinically appropriate.
Dendritic Cell Therapy for Triple-Negative Breast Cancer in Germany
Dendritic cell therapy for Triple-Negative Breast Cancer represents a personalized immune-based approach integrated into selected treatment plans. The process begins with comprehensive medical evaluation, including tumor profiling, laboratory assessment, and review by a multidisciplinary tumor board within structured oncology programs in Germany.
Once eligibility is confirmed, the procedure starts with collection of immune cells (monocytes) from the patient’s blood under controlled clinical conditions. These cells are transported to an EU GMP certified laboratory where they are processed under strict regulatory standards. In the laboratory, monocytes are differentiated and matured into functional dendritic cells capable of presenting tumor-associated antigens.
The prepared cells are then exposed to tumor-derived antigens specific to the patient’s cancer profile, resulting in a personalized dendritic cell vaccine. Prior to administration, quality control testing ensures sterility, viability, and biological activity. The vaccine is administered intradermally according to a defined schedule, followed by structured immune and clinical monitoring. The cost of Dendritic Cell Therapy in Germany is €27,000. This therapy is delivered within regulated programs, remains investigational, and is considered only in highly selected patients.
TACP Treatment for Triple-Negative Breast Cancer in Germany
TACP treatment for Triple-Negative Breast Cancer may be considered in selected patients with advanced or metastatic TNBC when dominant tumor sites require intensified regional chemotherapy delivery. This approach is particularly evaluated when certain metastatic deposits contribute significantly to disease progression or symptoms and when localized drug concentration may provide therapeutic benefit. TACP is designed to administer chemotherapy directly into the arteries supplying tumor tissue, thereby increasing local exposure while reducing overall systemic toxicity.
The procedure is performed under continuous real-time imaging guidance in a specialized interventional oncology unit. It starts with detailed radiologic assessment to identify the vascular supply of the targeted metastatic lesions and confirm technical suitability. A thin catheter is then inserted through a small arterial access point, typically in the groin, and carefully guided toward the vessels feeding the tumor. After precise positioning is confirmed with contrast imaging, chemotherapy is infused slowly and selectively into the tumor-supplying arteries to maximize cytotoxic concentration at the disease site.
Following completion of the infusion, the catheter is removed and the access site is secured. Patients are monitored for short-term effects and undergo scheduled follow-up imaging to assess treatment response and determine whether additional sessions are appropriate. Integration with systemic chemotherapy or immunotherapy is determined through multidisciplinary tumor board evaluation based on overall disease distribution and prior treatment history. The cost of TACP therapy in Germany typically ranges between €8,000 and €9,000 per session, depending on individualized clinical planning.
TACE Treatment for Triple-Negative Breast Cancer in Germany
TACE treatment for Triple-Negative Breast Cancer may be considered in selected patients when TNBC has spread to the liver and localized control of metastatic lesions is clinically necessary. In advanced TNBC, liver metastases can significantly influence overall disease progression and symptom burden, particularly when dominant tumor deposits affect organ function. TACE is designed to deliver concentrated chemotherapy directly into the blood vessels supplying liver metastases while simultaneously blocking their blood flow, thereby increasing local drug exposure and limiting systemic toxicity.
The procedure is performed under continuous real-time imaging guidance in a specialized interventional radiology setting. A catheter is inserted through a small arterial access point, usually in the groin, and carefully guided toward the vessels feeding the metastatic liver lesions. Once selective positioning is confirmed using contrast imaging, chemotherapy is infused directly into the targeted branches. This is followed by administration of embolic material that blocks the tumor blood supply, prolonging drug contact within cancer tissue and enhancing the localized therapeutic effect.
After TACE, patients undergo structured post-procedural monitoring, including evaluation of liver function and scheduled imaging reassessment to measure response. The treatment may be integrated with systemic chemotherapy or immunotherapy depending on overall disease distribution and multidisciplinary tumor board recommendations. The cost of TACE therapy in Germany typically ranges between €8,000 and €9,000 per session, depending on individualized planning and clinical considerations.
Ongoing Clinical Research and New Clinical Trials in Germany
German oncology institutions actively participate in research exploring new clinical trials for Triple-Negative Breast Cancer in Germany. Current studies investigate antibody-drug conjugates, combination immunotherapies, and cellular-based approaches aimed at improving outcomes in TNBC.
Research is conducted within regulated frameworks under Germany’s healthcare system, ensuring ethical oversight and patient safety. Collaboration with international organizations, including the American Cancer Society, supports scientific exchange and evidence-based development of treatment strategies.
Enrollment in clinical trials is reviewed by multidisciplinary tumor boards within recognized institutions considered among the best Triple-Negative Breast Cancer treatment centers. These programs aim to expand access to new and innovative treatment options for Triple-Negative Breast Cancer while maintaining responsible clinical standards.
Limitations and Considerations for Triple-Negative Breast Cancer
TNBC demonstrates biological variability that influences treatment response and long-term outcomes. While systemic chemotherapy, immunotherapy, and interventional approaches provide meaningful options, effectiveness depends on stage, molecular features, and overall patient health.
Interventional treatments such as TACE and TACP are appropriate for selected patients with dominant metastatic lesions and are not substitutes for systemic therapy in widespread disease. Similarly, dendritic cell therapy requires careful immune evaluation by the treating physician and structured follow-up within regulated programs.
Individualized planning through multidisciplinary tumor board evaluation remains essential. Ongoing clinical assessment and imaging surveillance support adaptive decision-making and balanced integration of advanced therapies into comprehensive cancer treatment strategies.
Why Choose Treatment for Triple-Negative Breast Cancer in Germany?
Cancer treatment in Germany is delivered through certified German healthcare centers operating under strict quality regulations. These institutions are recognized among the best Triple-Negative Breast Cancer treatment centers and integrate surgical oncology, systemic therapy, interventional procedures, and cellular immunotherapy within structured oncology programs in Germany.
Access to the latest medical technology in Germany supports precise diagnostics and minimally invasive procedures. Multidisciplinary tumor board evaluation ensures each patient receives a personalized and evidence-based treatment plan.
For international patients, treatment for Triple-Negative Breast Cancer in Germany provides coordinated care pathways supported by experienced oncology specialists and transparent clinical planning.
International Patient Support through Treatment in Germany (TIG)
Access to advanced management for Triple-Negative Breast Cancer involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as Immunotherapy Dendritic cell, TACP therapy and TACE therapy, through treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This preliminary evaluation by the treating physician helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
References
1- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020. CA Cancer J Clin. 2021;71(3):209–249. Open Access.
2- Bianchini G, Balko JM, Mayer IA, et al. Triple-negative breast cancer: challenges and opportunities. Nat Rev Clin Oncol. 2016;13(11):674–690. PMC Free Article.
3- National Cancer Institute. SEER Cancer Stat Facts: Female Breast Cancer. Public Registry.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:














.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)