Learn about stage 4 ovarian cancer treatment options including chemotherapy, immunotherapy, targeted therapies, and advanced interventional approaches available through specialized oncology centers in Germany.
What Are the Treatments for Stage 4 Ovarian Cancer?
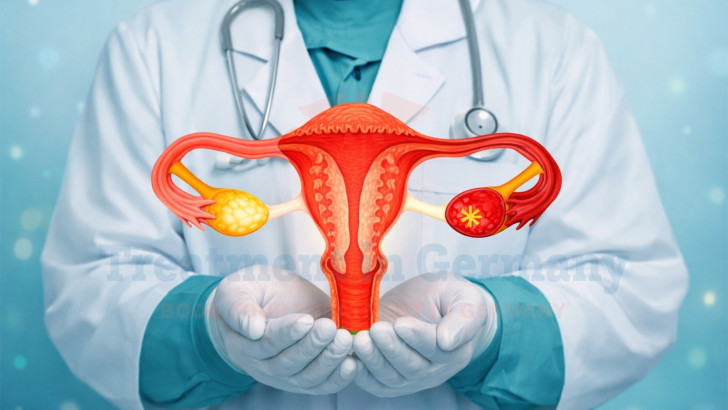
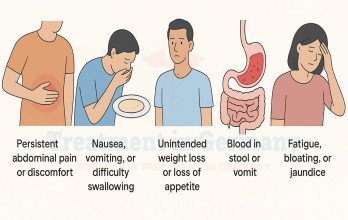
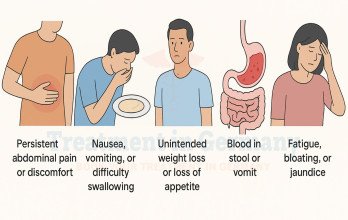
Ovarian cancer is a malignant tumor arising from the epithelial surface of the ovary, fallopian tube, or peritoneum. It is often referred to as a silent disease because early symptoms are vague and nonspecific, leading to delayed diagnosis in many patients. By the time ovarian cancer is identified, a substantial proportion of women already have advanced-stage disease involving distant organs or widespread peritoneal dissemination.
According to Global Cancer Statistics 2020, ovarian cancer accounted for 313,959 new cases (3.4%) and 207,252 deaths (4.7%) worldwide in 2020 [1]. These figures demonstrate the significant global burden of the disease. The SEER Cancer Stat Facts report that the 5-year relative survival rate for women with distant-stage ovarian cancer is 31%, based on cases diagnosed from 2014 through 2020 [2]. These data emphasize the clinical seriousness of stage 4 ovarian cancer treatment and the need for comprehensive therapeutic planning.
Stage IV ovarian cancer indicates that the malignancy has spread beyond the abdominal cavity to distant organs such as the liver parenchyma, lungs, or distant lymph nodes. Management requires multidisciplinary evaluation integrating surgical oncology, medical oncology, interventional radiology, and immune-based strategies. In leading German oncology centers, complex metastatic cases are reviewed in multidisciplinary tumor boards. Prof. Vogl is recognized as an expert in image-guided interventional oncology and embolization therapies, while Prof. Gansauge specializes in regulated cellular immunotherapy programs, including Immunotherapy Dendritic Cell Therapy. This coordinated framework within Germany’s healthcare system enables access to specialized ovarian cancer treatment centers, guided by experienced oncology specialists based on subspecialty expertise.
Understanding How Stage 4 Ovarian Cancer Develops and Progresses
Stage IV ovarian cancer represents distant metastatic disease. Tumor cells may spread through peritoneal fluid circulation, lymphatic dissemination, or hematogenous routes. While most ovarian cancers initially remain within the peritoneal cavity, advanced cases can involve the liver parenchyma, spleen, pleura, or lungs.
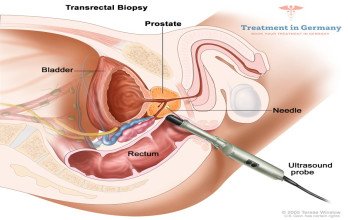
Accurate staging requires advanced diagnostics, including contrast-enhanced CT scans, MRI, and PET scans to evaluate the extent of disease. Imaging determines tumor burden, organ involvement, and suitability for surgical or interventional approaches. In selected cases, biopsy confirmation of distant lesions is required to establish definitive staging. Biologically, high-grade serous ovarian carcinoma is the most common histological subtype in advanced disease. Tumor cells demonstrate genomic instability, high proliferative activity, and interactions with the tumor microenvironment that promote dissemination. Comprehensive cancer treatment planning must consider tumor biology, metastatic distribution, and overall patient performance status.
Targeted Embolization Treatment for Stage 4 Ovarian Cancer in Germany
Targeted Embolization Treatment is an interventional oncology procedure designed to selectively block the blood vessels supplying metastatic tumor tissue. In Stage IV ovarian cancer, this approach may be evaluated when there are dominant metastatic lesions in organs such as the liver or pelvis that contribute significantly to symptom burden or disease progression.
The procedure is performed under image guidance using minimally invasive vascular access. A microcatheter is advanced into the specific arterial branches supplying the tumor. Once selective positioning is confirmed, embolic agents are delivered to block the blood flow to the tumor tissue. By decreasing vascular supply, embolization induces localized ischemia and promotes tumor necrosis.
In certain clinical situations, embolization may be combined with localized chemotherapy infusion to enhance cytotoxic concentration within metastatic deposits. Detailed pre-procedural imaging is essential to assess lesion size, vascular anatomy, and organ function. Multidisciplinary tumor board evaluation ensures appropriate patient selection within structured oncology programs.
Post-procedural monitoring includes observation for transient symptoms such as mild pain or fatigue. Follow-up imaging assesses radiologic response and determines whether additional sessions are indicated. As part of transparent treatment planning, financial aspects are clearly discussed, and the cost of TACE therapy in Germany typically ranges between €8,000 and €9,000 per session depending on clinical complexity and individualized planning requirements. In Stage IV ovarian cancer, this approach may be considered in selected patients with dominant metastatic lesions in organs such as the liver or pelvis, particularly when these sites contribute significantly to symptom burden or disease progression and after multidisciplinary evaluation.
Immunotherapy Dendritic Cell Therapy for Stage 4 Ovarian Cancer in Germany
Immunotherapy Dendritic Cell Therapy represents a personalized immune activation strategy integrated into advanced metastatic ovarian cancer management in selected patients. The process begins with comprehensive medical evaluation assessing immune competence, tumor burden, prior therapies, and organ function. Multidisciplinary tumor board discussion determines suitability within comprehensive cancer treatment planning.
Once eligibility is confirmed, the treatment begins with the collection of immune cells (monocytes) from the patient’s blood under medical supervision. These cells are transferred to an EU GMP certified laboratory, where strict regulatory standards govern processing, differentiation, and quality control. Within controlled laboratory conditions, monocytes are cultured and differentiated into mature dendritic cells capable of presenting tumor-associated antigens.
During preparation, dendritic cells are exposed to tumor-derived antigens specific to the patient’s cancer profile, resulting in a personalized dendritic cell vaccine. Before administration, the cellular product undergoes sterility testing, viability assessment, and identity verification. Administration is typically performed intradermally according to an individualized schedule, followed by clinical and immune monitoring. This regulated cellular therapy is delivered within structured programs in leading German oncology centers. As part of transparent planning, the cost of Immunotherapy Dendritic Cell Therapy is €27,000 which includes laboratory processing and vaccine preparation. This cellular immunotherapy remains investigational and is considered in highly selected patients within regulated clinical programs.
Ongoing Clinical Research and New Clinical Trials for Ovarian Cancer in Germany
Research efforts continue to explore antibody-drug conjugates, PARP inhibitor combinations, immune checkpoint inhibitors, and cellular immunotherapy approaches. Access to new clinical trials for ovarian cancer in Germany may be available for eligible patients following strict inclusion criteria.
Clinical research within Germany’s healthcare system aims to expand advanced ovarian cancer treatment in Germany while ensuring patient safety. Participation requires multidisciplinary evaluation and adherence to regulated protocols.
Limitations and Considerations in Advanced Stage 4 Ovarian Cancer
Therapeutic outcomes in stage 4 ovarian cancer are influenced by multiple factors, including tumor biology, the extent and distribution of metastases, prior treatment history, and the patient’s overall health status. Individualized treatment planning allows therapies to be adapted according to these variables, ensuring that each approach aligns with the patient’s clinical condition and goals of care. When carefully selected, Targeted embolization Treatment can contribute to meaningful local tumor control, symptom relief, and improved tolerability of systemic therapy as part of an integrated strategy.
Similarly, Immunotherapy Dendritic Cell Therapy represents a personalized immune-based approach that aims to stimulate tumor-specific immune responses. Its effectiveness may vary based on immune competence and tumor characteristics, which is why structured evaluation by the treating physician is essential before initiation. Multidisciplinary tumor board discussions within leading ovarian cancer treatment centers help determine optimal timing and integration of advanced therapies, supporting balanced decision-making and comprehensive cancer treatment planning tailored to the individual patient.
Why Choose Treatment in Germany for Stage 4 Ovarian Cancer?
Cancer treatment in Germany is supported by regulated clinical programs, precision-guided interventional infrastructure, and access to the latest medical technology in Germany. Leading institutions recognized among the leading ovarian cancer treatment centers integrate surgical oncology, systemic therapy, interventional radiology, and immune-based approaches.
Experienced German specialists collaborate within multidisciplinary tumor boards to provide individualized care pathways. For International patients, this structured coordination offers clarity and comprehensive support.
International Patient Support through Treatment in Germany (TIG)
Access to advanced management for ovarian cancer involves more than clinical decision-making; it requires careful logistical coordination, especially for international patients seeking specialized therapies such as Immunotherapy Dendritic cell therapy and Targeted Embolization Treatment, through treatment in Germany (TIG), medical documentation is reviewed in advance to facilitate appropriate specialist referral and structured treatment planning within experienced oncology centers. This preliminary evaluation by the treating physician helps align the patient’s clinical condition with available therapeutic options before travel arrangements are finalized.
Treatment in Germany (TIG) at www.treatmentingermany.de provides comprehensive logistical assistance, including hospital coordination, appointment scheduling, travel planning, and medical visa support when required. By organizing these administrative components in advance, patients can focus on clinical evaluation and multidisciplinary consultation upon arrival. Individuals considering advanced and innovative treatment approaches may get a free consultation with TIG (Treatment in Germany) to receive structured guidance and coordinated access to specialized cancer programs in Germany.
References
1- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020. CA Cancer J Clin. 2021;71(3):209–249. doi:10.3322/caac.21660. Open Access.
2- SEER Cancer Stat Facts: Ovarian Cancer. National Cancer Institute. Public Registry.
🌍Why Patients Worldwide Prefer Our Medical Services in Germany – Key Benefits Explained:














.webp)
 (1).webp)

.webp)
 (1).webp)

.jpg)